Chapter 8: Q75E (page 344)
Using to eliminate L - S. as wellas , obtain equation (8- 32 )from the equation that precedes it.
Short Answer
answer is
Learning Materials
Features
Discover
Chapter 8: Q75E (page 344)
Using to eliminate L - S. as wellas , obtain equation (8- 32 )from the equation that precedes it.
answer is
All the tools & learning materials you need for study success - in one app.
Get started for free
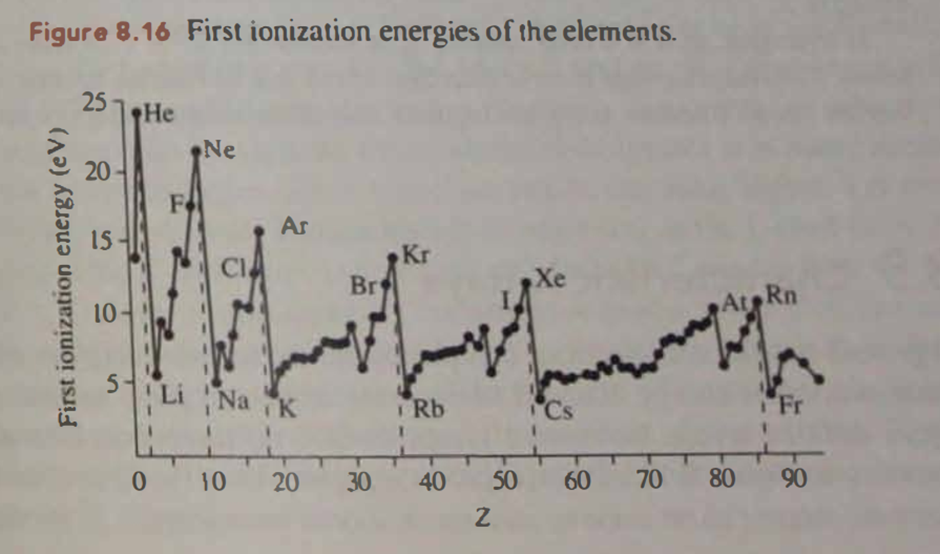
Question: Early on, the lanthanides were found to be quite uncooperative when attempts were made to chemically separate them from one another. One reason can be seen in Figure 8.16. Explain.
Question: The “radius of an atom” is a debatable quantity. Why?
Assume that the spin-orbit interaction is not overwhelmed by an external magnetic field what isthe minimum angle the total angular momentum vector may make with the z -axis in a3state of hydrogen?
In its ground state, carbon's 2pelectrons interact to produce . Given Hund's rule. what does this say about the total orbital angular momentum of these electrons?
Two particles in a box have a total energy .
(a) Which states are occupied?
(b) Make a sketch ofversusfor points along the line.
(c) Make a similar sketch of.
(d) Repeat parts (b) and (c) but for points on the line
What do you think about this solution?
We value your feedback to improve our textbook solutions.