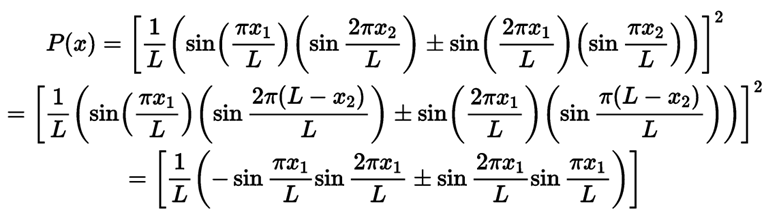Chapter 8: Q38E (page 340)
Two particles in a box have a total energy .
(a) Which states are occupied?
(b) Make a sketch ofversusfor points along the line.
(c) Make a similar sketch of.
(d) Repeat parts (b) and (c) but for points on the line
Short Answer
(a) The state of one particle is and other particles is
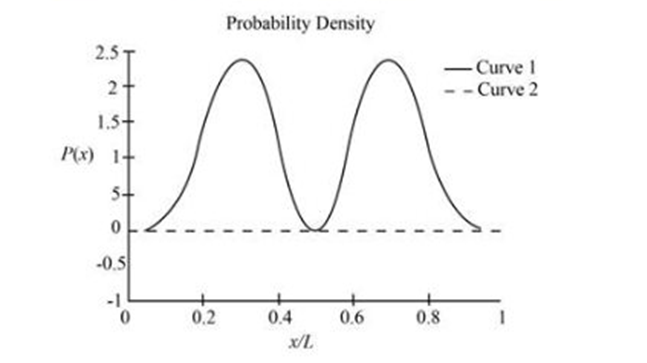
(b) The sketch is shown in figure 1
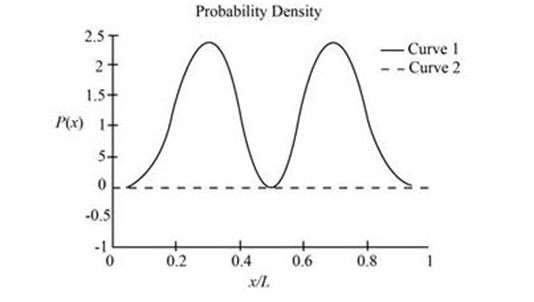
(c) The sketch is shown in figure 2
(d) The sketch is shown in figure 1 with curve B for symmetric function and curve A for asymmetric function.