Chapter 8: Q31E (page 339)
The subatomic omega particle has spin . What angles might its intrinsic angular in momentum vector make with the z-axis?
Short Answer
Angles that intrinsic angular momentum vector
Learning Materials
Features
Discover
Chapter 8: Q31E (page 339)
The subatomic omega particle has spin . What angles might its intrinsic angular in momentum vector make with the z-axis?
Angles that intrinsic angular momentum vector
All the tools & learning materials you need for study success - in one app.
Get started for free
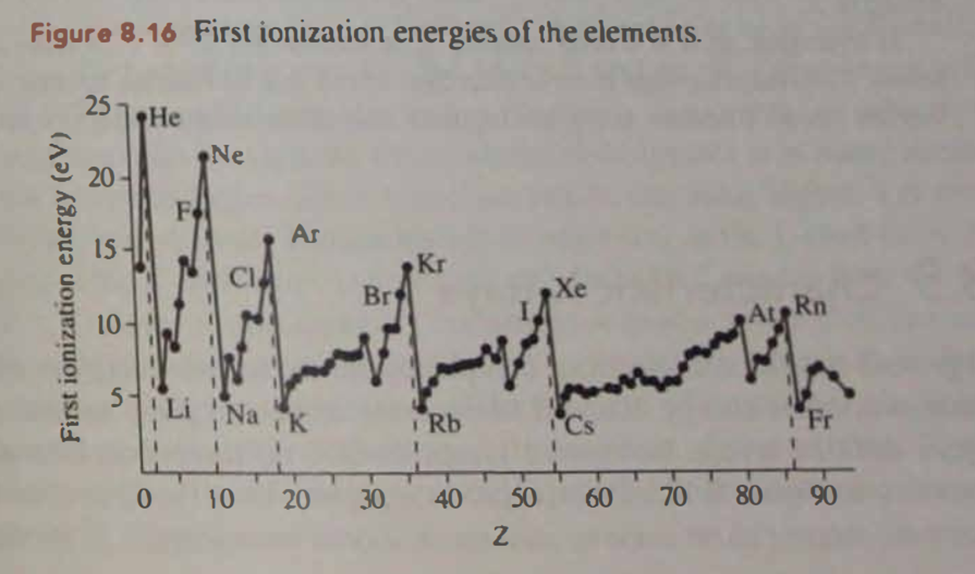
Question: As the 2p levels fill from Z = 5 to 10 the element's first ionization energies tend to increase. But as the figure below clearly indicates it is slightly easier to remove an electron from Z = 8 oxygen than from Z = 7 nitrogen. What might explain this?
The electron is known to have a radius no larger than . If actually produced by circulating mass, its intrinsic angular momentum of roughlywould imply very high speed, even if all that mass were as far from the axis as possible.
(a) Using simply(from |r × p|) for the angular momentum of a mass at radius r, obtain a rough value of p and show that it would imply a highly relativistic speed.
(b) At such speeds,andcombine to give(just as for the speedy photon). How does this energy compare with the known internal energy of the electron?
Question: The “radius of an atom” is a debatable quantity. Why?
Determine the electronic configuration for phosphorus, germanium and cesium.
In its ground state, carbon's 2pelectrons interact to produce . Given Hund's rule. what does this say about the total orbital angular momentum of these electrons?
What do you think about this solution?
We value your feedback to improve our textbook solutions.