Chapter 8: Q22CQ (page 339)
Question: Huge tables of characteristic X-rays start at lithium. Why not hydrogen or helium?
Short Answer
Answer
X-rays do not start at hydrogen or helium because all of the electrons are n= 1 electrons.
Learning Materials
Features
Discover
Chapter 8: Q22CQ (page 339)
Question: Huge tables of characteristic X-rays start at lithium. Why not hydrogen or helium?
Answer
X-rays do not start at hydrogen or helium because all of the electrons are n= 1 electrons.
All the tools & learning materials you need for study success - in one app.
Get started for free
Angular momenta and interact so that they obey the strict quantum mechanical rules for angular momentum addition. If and what angles between and allowed?
Question:Figure 8.16 shows that in the Z = 3 to 10 filling of the n = 2 shell (lithium to neon), there is an upward trend in elements' first Ionization energies. Why is there a drop as Z goes from 4 to 5, from beryllium to boron?
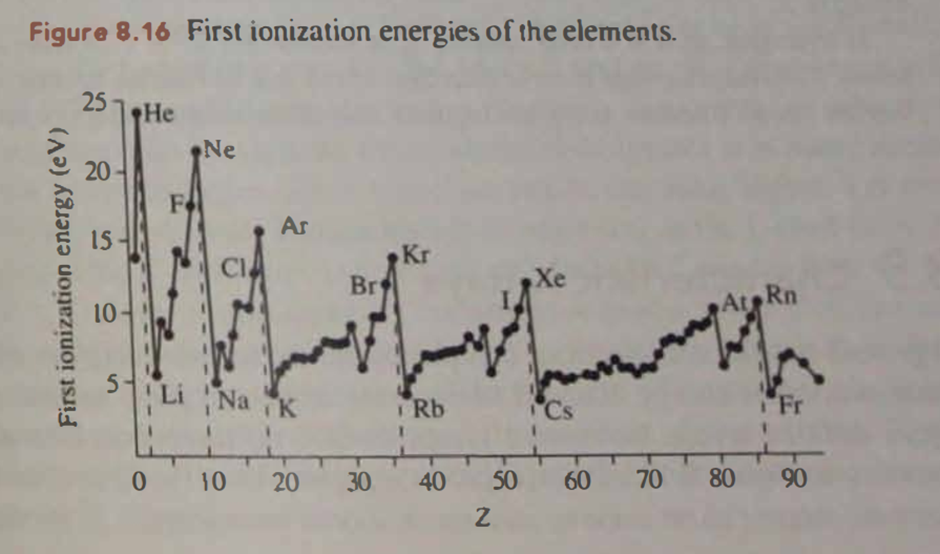
Question: Solving (or attempting to solve!) a 4-electron problem is not twice as hard as solving a 2-electrons problem. Would you guess it to be more or less than twice as hard? Why?
What is the minimum possible energy for five (non-interacting) spin particles of massmin a one dimensional box of length L ? What if the particles were spin-1? What if the particles were spin ?
Summarize the connection between angular momentum quantization and the stem-Gerlach experiment.
What do you think about this solution?
We value your feedback to improve our textbook solutions.