Chapter 8: 70E (page 344)
Angular momenta and interact so that they obey the strict quantum mechanical rules for angular momentum addition. If and what angles between and allowed?
Short Answer
The first angle allowed between J1 and J2 is .
The second angle allowed between J1 and J2 is .
The last angle allowed between J1 and J2 is .
Step by step solution
Given data
Given data is and .
Formula of angular moment.
In order to find the angle between the quantum mechanical angular momentum vectors J1 and J2 when the quantum numbers for me two of them are 1 and 3/2 respectively, equations for the angular momentum, the quantum number for the angular momentum, and the law of cosines will be needed.
The magnitude of some angular momentum J.
……. (1)
Here data-custom-editor="chemistry" ¡s Planck's reduced constant, and data-custom-editor="chemistry" is the quantum number for some angular momentum.
The quantum number for the total angular momentumdata-custom-editor="chemistry" .
data-custom-editor="chemistry" ……. (2)
Here j1 and j2 are the quantum numbers for angular momentum vectors J1 and J2 respectively.
The law of cosines relating the lengths of sides a, b and c and angle data-custom-editor="chemistry" between sides a and b is, data-custom-editor="chemistry" .
Find the value of φ
The general picture for the relation between and data-custom-editor="chemistry" looks like in figure 1. 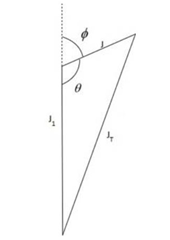
Figure 1
Here, angle between J1 and J2 is data-custom-editor="chemistry" .
Use law of cosine and magnitudes of the vectors.
Apply angle data-custom-editor="chemistry" instead of data-custom-editor="chemistry" .
From the diagram, data-custom-editor="chemistry" , solve for data-custom-editor="chemistry" .
role="math" localid="1658395808801" ……. (3)
Find the magnitude of JT
Use this, we must know magnitudes of vectors J1 , J2 and JT which can be obtained in quantum numbers j1, j2 and jT.
j1 And j2 are 1 and 3/2 respectively, so those can be inserted into equation (1) for the magnitudes of J1 and J2.
Find the magnitude of data-custom-editor="chemistry" , you need to know what the quantum numbers for that are, use equation (2), with 1 for j1 and 3/2 for j2.
data-custom-editor="chemistry"
Simplify further as shown below.
Given that 1 is added in increments to data-custom-editor="chemistry" until you reach (according to equation (2)), all the values for data-custom-editor="chemistry" .
data-custom-editor="chemistry"
So the equation (1) can be used to find the values for the various data-custom-editor="chemistry" .
role="math" localid="1658397950336"
Find the first allowed angle between J1 and J2
Find the first angle allowed between J1 and J2 , is used for J1, is used for J2 and is used for JT in equation (3).
So the first angle allowed between J1 and J2 is role="math" localid="1658399171396" .
Find the second allowed angle between J1 and J2
Find the second angle allowed between J1 and J2 , is used for J1, is used for J2 and is used for JT in equation (3).
So the second angle allowed between J1 and J2 is .
Find the last allowed angle between J1 and J2
Find the third angle allowed between J1 and J2, data-custom-editor="chemistry" is used for J1, is used for J2 , and is used for JT in equation (3).
role="math" localid="1658400686867"
So the last angle allowed between J1 and J2 is 56.8o.
The first angle allowed between J1 and J2 is 156o.
The second angle allowed between J1 and J2 is 111o.
The last angle allowed between J1 and J2 is 56.8o.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!