Chapter 10: Q67E (page 470)
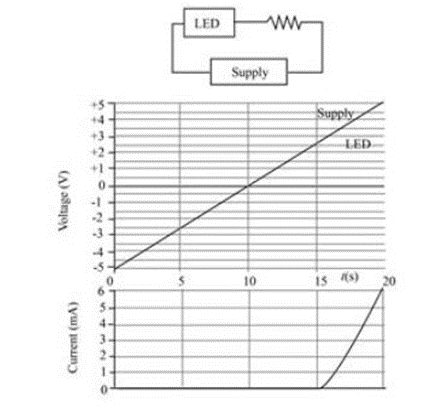
Question: An LED is connected in series with a resistor and a power supply of variable voltage. The supply voltage starts from —5 V and increases linearly with time to a maximum of +5 V. The figure shows the supply voltage the voltage across the diode, and the current in the Circuit as functions of time. The LED does not glow for the whole time shown. (a) Explain the plots, and identify the period when the LED glows. (b) Approximately what color is the light produced by the LED?
Short Answer
Answer
(a) The band gap of the LED is 2.5e V .
(b) The light produced by the LED is cyan color light.