Chapter 10: 2CQ (page 413)
Upon what definitions do we base the claim that the and states of equations are related to x and y just as is to .
Short Answer
It is proven by converting Cartesian to spherical polar coordinates.
Learning Materials
Features
Discover
Chapter 10: 2CQ (page 413)
Upon what definitions do we base the claim that the and states of equations are related to x and y just as is to .
It is proven by converting Cartesian to spherical polar coordinates.
All the tools & learning materials you need for study success - in one app.
Get started for free
Describe the similarities and differences between Type-I and Type-II superconductors.
Question: - (a) Compare equation (10-11) evaluated at room temperature for a silicon band gap of 1.1 eV and for a typical donor-state/conduction band gap of 0.05 eV.
(b) Assuming only one impurity atom for every 10³ silicon atoms, do your results suggest that majority carriers, bumped up from donor levels. should outnumber minority carriers created by thermal excitation across the whole 1.1 eV gap? (The calculation ignores the difference in density of states between donor levels and bands, which actually strengthens the argument.)
The vertices of a tetrahedron are four vertices of a cube symmetrically chosen so that no two are adjacent. Show that the angle between the vertices of a tetrahedron is .
For the four kinds of crystal binding – covalent, ionic, metallic, and molecular- how would the destiny of valence electrons vary throughout the solid? Would it be constant, centered on the atoms, or largest between the atoms? Or would it alternate, with a net charge density positive at one atom and negative at the next?
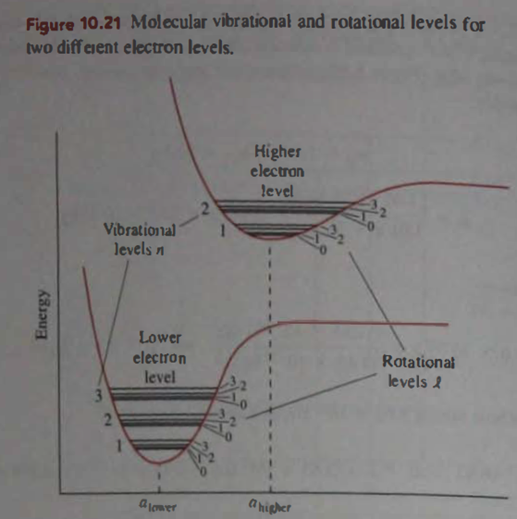
Question: From the qualitative shapes of the interatomic potential energies in Fig. 10.21, would you expect the vibrational level in the excited electronic state to be spaced the same. Farther apart, or closer together than those in the lower energy electronic state? Explain what about the rotational levels?
What do you think about this solution?
We value your feedback to improve our textbook solutions.