Chapter 10: Q43E (page 469)
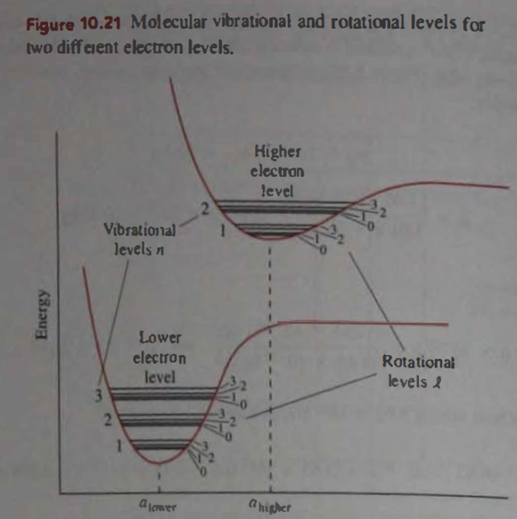
Question: From the qualitative shapes of the interatomic potential energies in Fig. 10.21, would you expect the vibrational level in the excited electronic state to be spaced the same. Farther apart, or closer together than those in the lower energy electronic state? Explain what about the rotational levels?
Short Answer
Answer
- Due to the smaller value of fraction, the vibrational levels will be closely packed. As The minima for rotational levels are located at larger separation, thus, the levels are more closely packed due to increased rotational inertia


