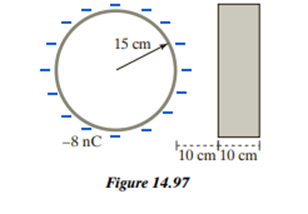
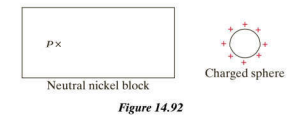
Chapter 14: Q2CP (page 552)
A student asked, "Since the positive nucleus of the atom is hidden inside a negative electron cloud, why doesn't all matter appear to be negatively charged?" Explain to the student the flaw in this reasoning.
Short Answer
The atom becomes neutral because of the polarization of both the positive and the negative charges. The electron is centrally located in the nucleus. However, if the electron is centrally positioned, the proton field will be canceled by the electrons and it will create its own field. Thus, the atom appears as a neutral atom rather than a negatively charged atom.