Chapter 18: Q3P (page 541)
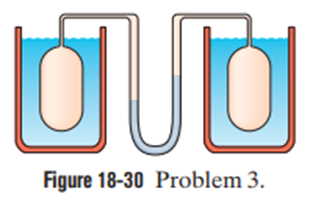
Question: A gas thermometer is constructed of two gas-containing bulbs, each in a water bath, as shown in Figure. The pressure difference between the two bulbs is measured by a mercury manometer as shown. Appropriate reservoirs, not shown in the diagram, maintain constant gas volume in the two bulbs. There is no difference in pressure when both baths are at the triple point of water. The pressure difference is 120 torr when one bath is at the triple point and the other is at the boiling point of water. It is 90.0 torrwhen one bath is at the triple point and the other is at an unknown temperature to be measured. What is the unknown temperature?
Short Answer
Answer:
The unknown temperature is 348 K



