Chapter 38: Q46P (page 1183)
Calculate the de Broglie wavelength of (a) a electron, (b) a photon, and (c) aneutron.
Short Answer
(a) The wavelength of an electron is .
(b) The wavelength of the photon is .
(c) The wavelength of the neutron is .
Learning Materials
Features
Discover
Chapter 38: Q46P (page 1183)
Calculate the de Broglie wavelength of (a) a electron, (b) a photon, and (c) aneutron.
(a) The wavelength of an electron is .
(b) The wavelength of the photon is .
(c) The wavelength of the neutron is .
All the tools & learning materials you need for study success - in one app.
Get started for free
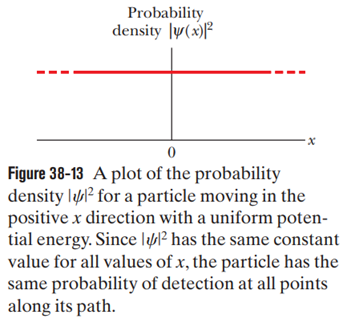
Suppose we put in Eq. 38-24 and relabeled as localid="1664290358337" .
(a) What would the resulting wave function then describe?
(b) How, if at all, would Fig. 38-13 be altered?
Find the maximum kinetic energy of electrons ejected from a certain material if the material’s work function is 2.3 eV and the frequency of the incident radiation is .
Imagine playing baseball in a universe (not ours!) where the Planck constant is 0.60 J.s, and thus quantum physics affects macroscopic objects. What would be the uncertainty in the position of a 0.50 kg baseball that is moving at 20 m/s along an axis if the uncertainty in the speed is 1.0 m/s?
The existence of the atomic nucleus was discovered in by Ernest Rutherford, who properly interpreted some experiments in which a beam of alpha particles was scattered from a metal foil of atoms such as gold. (a) If the alpha particles had a kinetic energy of , what wads their de Broglie wavelength? (b) Explain whether the wave nature of the incident alpha particles should have been taken into account in interpreting these experiments. The mass of an alpha particle is (atomic mass units), and its distance of closest approach to the nuclear center in these experiments was about . (The wave nature of matter was not postulated until more than a decade after these crucial experiments were performed.)
(a) In Me V/c, what is the magnitude of momentum associated with a photon having an energy equal to the electron rest energy? What are the (b) wavelength and (c) frequency of the corresponding radiation?
What do you think about this solution?
We value your feedback to improve our textbook solutions.