Chapter 38: Q14Q (page 1181)
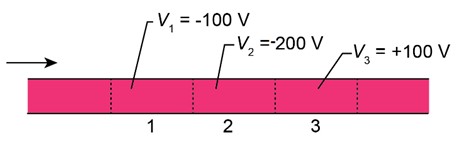
Figure 38-24 shows an electron moving through several regions where uniform electric potentials V have been set up. Rank the three regions according to the de Broglie wavelength of the electron there, greatest first.
Short Answer
The rank is.

