Chapter 39: Q64P (page 1218)
Verify that the combined value of the constants appearing in Eq. 39-33 is 13.6eV
Short Answer
It is verify that the combined value of the constants appearing in Eq. 39-33 is 13.6 eV.
Learning Materials
Features
Discover
Chapter 39: Q64P (page 1218)
Verify that the combined value of the constants appearing in Eq. 39-33 is 13.6eV
It is verify that the combined value of the constants appearing in Eq. 39-33 is 13.6 eV.
All the tools & learning materials you need for study success - in one app.
Get started for free
A proton is confined to a one-dimensional infinite potential well 100pm wide. What is its ground-state energy?
A diatomic gas molecule consists of two atoms of massseparated by a fixed distance drotating about an axis as indicated in given figure. Assuming that its angular momentum is quantized as in the Bohr model for the hydrogen atom, find
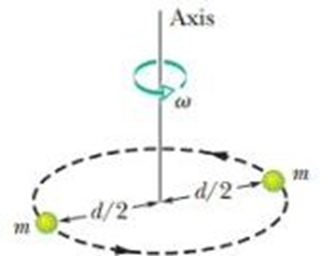
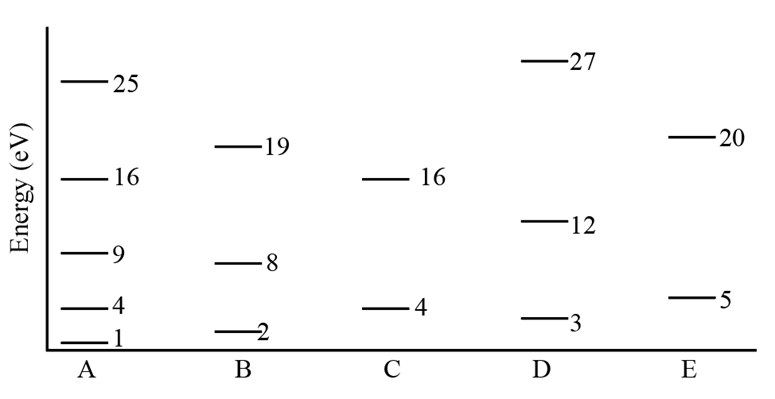
Figure 39-26 indicates the lowest energy levels (in electronvolts) for five situations in which an electron is trapped in a one-dimensional infinite potential well. In wells B, C, D, and E, the electron is in the ground state. We shall excite the electron in well A to the fourth excited state (at 25 eV). The electron can then de-excite to the ground state by emitting one or more photons, corresponding to one long jump or several short jumps. Which photon emission energies of this de-excitation match a photon absorption energy (from the ground state) of the other four electrons? Give then values.
An electron is contained in the rectangular box of Fig. 39-14, with widths , , and . What is the electron’s ground-state energy?
Calculate the radial probability density P(r) for the hydrogen atom in its ground state at (a) r = 0 , (b) r = a , and (c) r = 2a, where a is the Bohr radius.
What do you think about this solution?
We value your feedback to improve our textbook solutions.