Chapter 20: Q45P (page 606)
Construct a table like Table20 - 1for eight molecules.
Short Answer
The table for eight molecules is constructed.
Learning Materials
Features
Discover
Chapter 20: Q45P (page 606)
Construct a table like Table20 - 1for eight molecules.
The table for eight molecules is constructed.
All the tools & learning materials you need for study success - in one app.
Get started for free
A box contains Nmolecules. Consider two configurations: Configuration Awith an equal division of the molecules between the two halves of the box, and configuration Bwith 60.0%of the molecules in the left half of the box and 40.0%in the right half. For N =50, what are (a) the multiplicity WAof configuration A, (b) the multiplicity of configuration B, and (c) the ratioof the time the system spends in configuration Bto the time it spends in configuration A? For N =100, what are (d), (e), and (f)? ForN =200, what are (g), (h), and (i)? ( j) With increasingN, doesincrease, decrease, or remain the same?
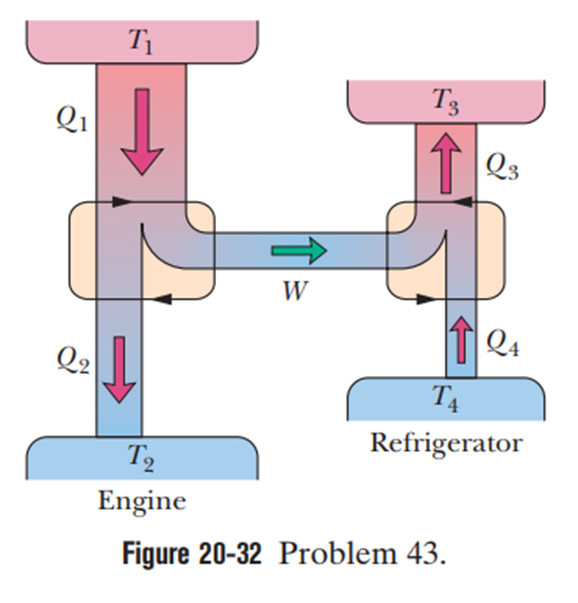
Figure 20-32 represents a Carnot engine that works between temperatures and and drives a Carnot refrigerator that works between temperatures . What is the ratio ?
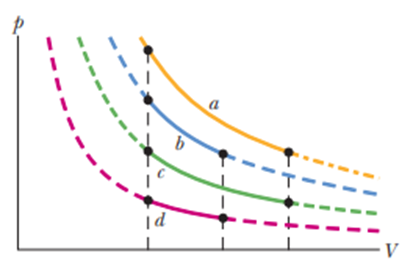
In four experiments, 2.5 molof hydrogen gas undergoes reversible isothermal expansions, starting from the same volume but at different temperatures. The corresponding P-Vplots are shown in Fig. 20-21. Rank the situations according to the change in the entropy of the gas, greatest first.
A mixture of1773g of water and 227gof ice is in an initial equilibrium state at . The mixture is then, in a reversible process, brought to a second equilibrium state where the water – ice ratio, by mass, is 1.00 : 1.00at. (a)Calculate the entropy change of the system during this process. (The heat of fusion for water is 333 kJ/kg.)(b) The system is then returned to the initial equilibrium state in an irreversible process (say, by using a Bunsen burner). Calculate the entropy change of the system during this process. (c)Are your answers consistent with the second law of thermodynamics?
(a) What is the entropy change of a 12.0gice cube that melts completely in a bucket of water whose temperature is just above the freezing point water?(b) What is the entropy change of a 5.00gspoonful of water that evaporates completely on a hot plate whose temperature is slightly above the boiling point of water?
What do you think about this solution?
We value your feedback to improve our textbook solutions.