Chapter 20: Q43P (page 606)
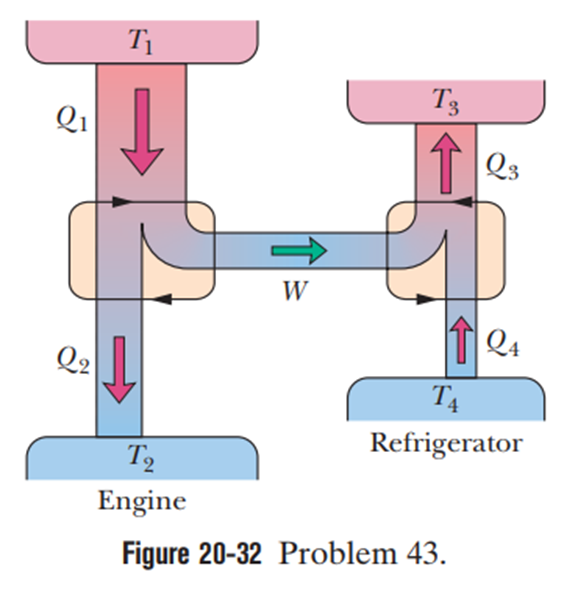
Figure 20-32 represents a Carnot engine that works between temperatures and and drives a Carnot refrigerator that works between temperatures . What is the ratio ?
Short Answer
The value for the ratio .
Learning Materials
Features
Discover
Chapter 20: Q43P (page 606)
Figure 20-32 represents a Carnot engine that works between temperatures and and drives a Carnot refrigerator that works between temperatures . What is the ratio ?

The value for the ratio .
All the tools & learning materials you need for study success - in one app.
Get started for free
A 2.50molsample of an ideal gas expands reversibly and isothermally atuntil its volume is doubled. What is the increase in entropy of the gas?
In four experiments, blocks Aand B, starting at different initialtemperatures, were brought together in an insulating box and allowedto reach a common final temperature. The entropy changesfor the blocks in the four experiments had the following values (injoules per Kelvin), but not necessarily in the order given.Determine which values for Ago with which values for B.
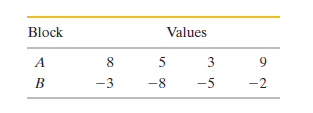
An ideal gas undergoes a reversible isothermal expansion at , increasing its volume fromto. The entropy change of the gas is. How many moles of gas are present?
Three Carnot engines operate between temperature limits of (a) 400 and 500K, (b) 500 and 600K, and (c) 400 and 600K. Each engine extracts the same amount of energy per cycle from the high-temperature reservoir. Rank the magnitudes of the work done by the engines per cycle, greatest first.
Expand 1.00 molof a monatomic gas initially at 5.00kPaand 600 Kfrom initial volumeto final volume. At any instant during the expansion, the pressure pand volume Vof the gas are related by, with pin kilopascals, and Vin cubic meters, and. (a) What is the final pressure and (b) what is the final temperature of the gas? (c) How much work is done by the gas during the expansion? (d) What isfor the expansion? (Hint: Use two simple reversible processes to find.)
What do you think about this solution?
We value your feedback to improve our textbook solutions.