Chapter 20: Q3P (page 604)
A 2.50molsample of an ideal gas expands reversibly and isothermally atuntil its volume is doubled. What is the increase in entropy of the gas?
Short Answer
The increase in the entropy of the gas is 14.4J/K
Learning Materials
Features
Discover
Chapter 20: Q3P (page 604)
A 2.50molsample of an ideal gas expands reversibly and isothermally atuntil its volume is doubled. What is the increase in entropy of the gas?
The increase in the entropy of the gas is 14.4J/K
All the tools & learning materials you need for study success - in one app.
Get started for free
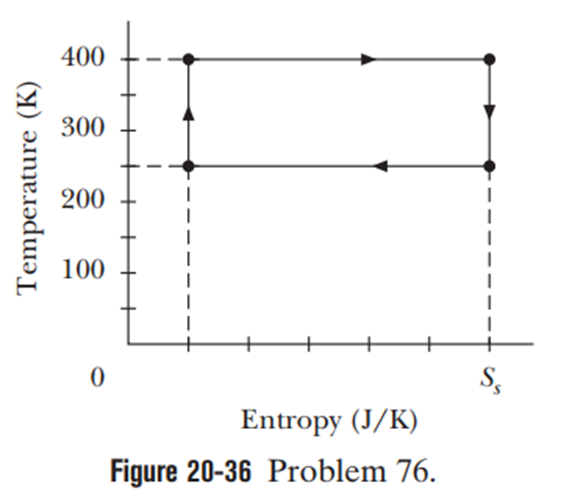
Figure 20-36 shows a Carnot cycle on a T-Sdiagram, with a scale set by. For a full cycle, find (a) the net heat transfer and (b) the net work done by the system.
Suppose that a deep shaft were drilled in Earth’s crust near one of the poles, where the surface temperature is , to a depth where the temperature is . (a) What is the theoretical limit to the efficiency of an engine operating between these temperatures? (b) If all the energy released as heat into the low-temperature reservoir were used to melt ice that was initially at , at what rate could liquid water at be produced by a 100 MW power plant (treat it as an engine)? The specific heat of ice is ; water’s heat of fusion is . (Note that the engine can operate only between and in this case. Energy exhausted atcannot warm anything above .)
A brass rod is in thermal contact with a constant-temperature reservoir atat one end and a constant-temperature reservoir at at the other end. (a) Compute the total change in entropy of the rod–reservoirs system when 5030 Jof energy is conducted through the rod, from one reservoir to the other. (b) Does the entropy of the rod change?
How much energy must be transferred as heat for a reversible isothermal expansion of an ideal gas at if the entropy of the gas increases by 46.0 J/K?
An ideal monatomic gas at initial temperature(in Kelvins) expands from initial volume to volumeby each of the five processes indicated in the T-Vdiagram of Fig. 20-20. In which process is the expansion (a) isothermal, (b) isobaric (constant pressure), and (c) adiabatic? Explain your answers. (d) In which processes does the entropy of the gas decrease?
What do you think about this solution?
We value your feedback to improve our textbook solutions.