For simplification, we redraw the figure.
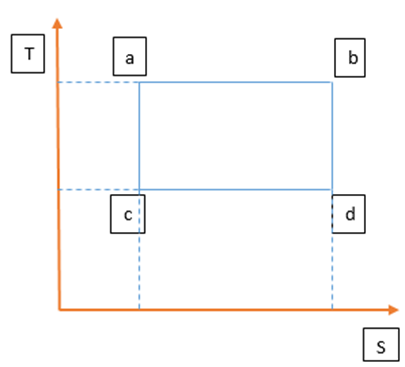
For paths ab and cd , the process is isothermal, so the temperature is constant. Let be the heat transformed at the path ab and be the heat transformed at the path cd . For paths ac and bc, the process is adiabatic, so the total heat is constant and entropy is also constant.
As entropy is set by the scale
The change in entropy for the pathis given as:
The net heat transformed at high temperature,i.e., is calculated by using equation (2) as:
role="math" localid="1661337709961"
The cycle is complete, so the total internal energy is constant.
Similarly, for path cd, the change in entropy is given as:
The net heat transformed at low temperature i.e.is calculated by using equation (2) as:
Now, the net heat transferred in the cycle is given as:
Hence, the value of the net energy transfer is 75 J



