Chapter 20: Q5Q (page 603)
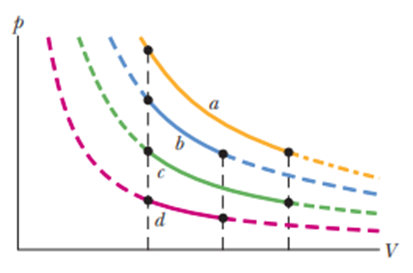
In four experiments, 2.5 molof hydrogen gas undergoes reversible isothermal expansions, starting from the same volume but at different temperatures. The corresponding P-Vplots are shown in Fig. 20-21. Rank the situations according to the change in the entropy of the gas, greatest first.
Short Answer
The ranking of the situations according to the change in entropy of the gas is


