Chapter 20: Q18P (page 605)
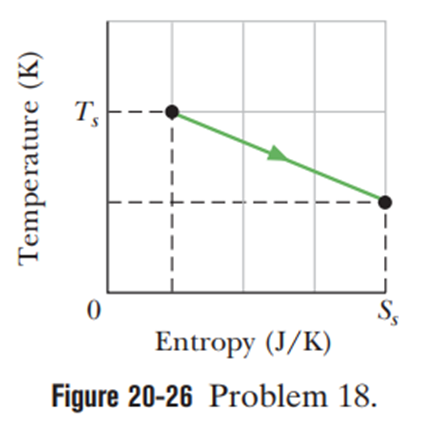
A 2.0 molsample of an ideal monatomic gas undergoes the reversible process shown in Figure. The scale of the vertical axis is set byand the scale of the horizontal axis is set by. (a) How much energy is absorbed as heat by the gas? (b) What is the change in the internal energy of the gas? (c) How much work is done by the gas?
Short Answer
a) Energy absorbed as heat by the gas is .
b) Change in internal energy of the gas is .
c) Work done by the gas is 9.5 kJ.