Chapter 20: Q17P (page 605)
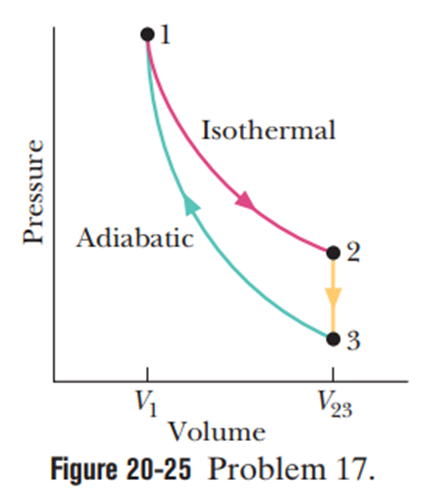
In Figure 20-25, where,n moles of a diatomic ideal gas are taken through the cycle with the molecules rotating but not oscillating. What are (a), (b), and (c)? For path, what are (d), (e), (f), and (g)? For path, what are (h),(i)(j), and (k)? For path, what are (l),(m)role="math" localid="1661575470031" (n), and (o)?
Short Answer
a) Ratio of is 0.333
b) Ratio of is 0.215
c) Ratio of is 0.644
d) For path ratio of is 1.10
e) For path , the ratio of is 1.10
f) For path ratio of is 0
g) For path , the ratio of is 1.10
h) For path ratio of is 0
i) For path , the ratio of is -0.889
j) For path ratio of is -0.889
k) For path , the ratio of is -1.10
l) For path ratio of is -0.889
m) For path , the ratio of is 0
n) For path ratio of is 0.889
o) For path , the ratio of is 0