Determine the constant in Eq. 40-27 to five significant figures by finding in terms of the fundamental constants in Eq. 40-24 and then using data from Appendix B to evaluate those constants. Using this value of in Eq. 40-27, determine the theoretical energy of the photon for the low-mass elements listed in the following table. The table includes the value (eV) of the measured energy of the photon for each listed element. The percentage deviation between and can be calculated as:
What is the percentage deviation for (a) Li, (b) Be, (c) B, (d) C, (e) N, (f) O, (g) F, (h) Ne, (i) Na, and (j) Mg?
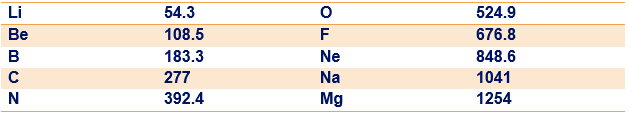
(There is actually more than one ray because of the splitting of the energy level, but that effect is negligible for the elements listed here.)



