Chapter 40: Q42P (page 1249)
From Fig. 40-13, calculate approximately the energy difference for molybdenum. Compare it with the value that may be obtained from Fig. 40-15.
Short Answer
The approximate energy differencefor molybdenum is 2.2 keV.
Learning Materials
Features
Discover
Chapter 40: Q42P (page 1249)
From Fig. 40-13, calculate approximately the energy difference for molybdenum. Compare it with the value that may be obtained from Fig. 40-15.
The approximate energy differencefor molybdenum is 2.2 keV.
All the tools & learning materials you need for study success - in one app.
Get started for free
Show that a moving electron cannot spontaneously change into an x-ray photon in free space. A third body (atom or nucleus) must be present. Why is it needed? (Hint: Examine the conservation of energy and momentum.)
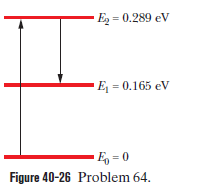
Martian laser. Where sunlight shines on the atmosphere of Mars, carbon dioxide molecules at an altitude of about 75 km undergo natural laser action. The energy levels involved in the action are shown in Fig. 40-26; population inversion occurs between energy levels and . (a) What wavelength of sunlight excites the molecules in the lasing action? (b) At what wavelength does lasing occur? (c) In what region of the electromagnetic spectrum do the excitation and lasing wavelengths lie?
A tungsten (Z=74) target is bombarded by electrons in an x-ray tube. The K,L and M energy levels for tungsten (compare Fig. 40-15) have the energies 69.5 keV,11.3 keV, and 2.30 keV respectively. (a) What is the minimum value of the accelerating potential that will permit the production of the characteristic and lines of tungsten? (b) For this same accelerating potential, what is ? What are the (c) and (d) wavelengths?
What is the acceleration of a silver atom as it passes through the deflecting magnet in the Stern–Gerlach experiment of Fig. 40-8 if the magnetic field gradient is 1.4 T/mm?
A laser emits at 424 nm in a single pulse that lasts .The power of the pulse is 2.80 MW . If we assume that the atoms contributing to the pulse underwent stimulated emission only once during the ,how many atoms contributed?
What do you think about this solution?
We value your feedback to improve our textbook solutions.