Chapter 40: Q64P (page 1250)
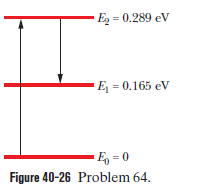
Martian laser. Where sunlight shines on the atmosphere of Mars, carbon dioxide molecules at an altitude of about 75 km undergo natural laser action. The energy levels involved in the action are shown in Fig. 40-26; population inversion occurs between energy levels and . (a) What wavelength of sunlight excites the molecules in the lasing action? (b) At what wavelength does lasing occur? (c) In what region of the electromagnetic spectrum do the excitation and lasing wavelengths lie?
Short Answer
(a) The wavelength of sunlight that excites the molecules in the lasing action is .
(b) The lasing occurs at wavelength of .
(c) The electromagnetic spectrum in which the excitation and the wavelengths lie is in infrared region.


