Chapter 40: Q77P (page 1251)
Knowing that the minimum x-ray wavelength produced by 40.0 keV electrons striking a target is 31.1 pm, determine the Planck constant .
Short Answer
The value of Planck’s constant h is .
Learning Materials
Features
Discover
Chapter 40: Q77P (page 1251)
Knowing that the minimum x-ray wavelength produced by 40.0 keV electrons striking a target is 31.1 pm, determine the Planck constant .
The value of Planck’s constant h is .
All the tools & learning materials you need for study success - in one app.
Get started for free
Here are the wavelengths of a few elements:
Element | λ(�賾) | Element | λ(�賾) |
Ti | 275 | Co | 179 |
V | 250 | Ni | 166 |
Cr | 229 | Cu | 154 |
Mn | 210 | Zn | 143 |
Fe | 193 | Ga | 134 |
Make a Moseley plot (like that in Fig. 40-16) from these data and verify that its slope agrees with the value given for C in Module 40-6.
Show that the number of states with the same quantum number nis .
Determine the constant in Eq. 40-27 to five significant figures by finding in terms of the fundamental constants in Eq. 40-24 and then using data from Appendix B to evaluate those constants. Using this value of in Eq. 40-27, determine the theoretical energy of the photon for the low-mass elements listed in the following table. The table includes the value (eV) of the measured energy of the photon for each listed element. The percentage deviation between and can be calculated as:
What is the percentage deviation for (a) Li, (b) Be, (c) B, (d) C, (e) N, (f) O, (g) F, (h) Ne, (i) Na, and (j) Mg?
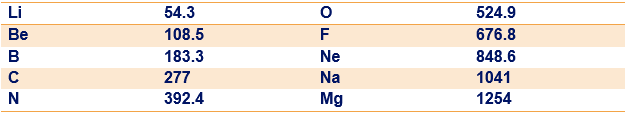
(There is actually more than one ray because of the splitting of the energy level, but that effect is negligible for the elements listed here.)
What is the acceleration of a silver atom as it passes through the deflecting magnet in the Stern–Gerlach experiment of Fig. 40-8 if the magnetic field gradient is 1.4 T/mm?
A recently named element is darmstadtium (Ds), which has electrons. Assume that you can put 110 the electrons into the atomic shells one by one and can neglect any electron-electron interaction. With the atom in the ground state, what is the spectroscopic notation for the quantum number for the last electron?
What do you think about this solution?
We value your feedback to improve our textbook solutions.