Chapter 2: Q. 2.32 (page 79)
Find an expression for the entropy of the two-dimensional ideal gas considered in Problem . Express your result in terms of ,and.
Short Answer
The Entropy of the two-dimensional ideal gaas is
Learning Materials
Features
Discover
Chapter 2: Q. 2.32 (page 79)
Find an expression for the entropy of the two-dimensional ideal gas considered in Problem . Express your result in terms of ,and.
The Entropy of the two-dimensional ideal gaas is
All the tools & learning materials you need for study success - in one app.
Get started for free
Use the methods of this section to derive a formula, similar to equation, for the multiplicity of an Einstein solid in the "low-temperature" limit, .
Rather than insisting that all the molecules be in the left half of a container, suppose we only require that they be in the leftmost (leaving the remaining completely empty). What is the probability of finding such an arrangement if there are molecules in the container? What if there are molecules? What if there are ?
For either a monatomic ideal gas or a high-temperature Einstein solid, the entropy is given by times some logarithm. The logarithm is never large, so if all you want is an order-of-magnitude estimate, you can neglect it and just say . That is, the entropy in fundamental units is of the order of the number of particles in the system. This conclusion turns out to be true for most systems (with some important exceptions at low temperatures where the particles are behaving in an orderly way). So just for fun, make a very rough estimate of the entropy of each of the following: this book (a kilogram of carbon compounds); a moose of water ; the sun of ionized hydrogen .
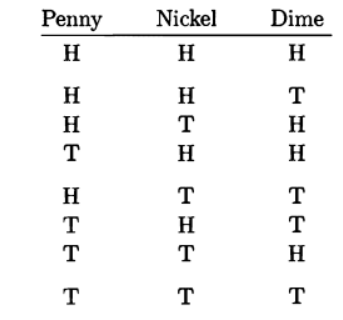
Suppose you flip four fair coins.
(a) Make a list of all the possible outcomes, as in Table 2.1.
(b) Make a list of all the different "macrostates" and their probabilities.
(c) Compute the multiplicity of each macrostate using the combinatorial formula , and check that these results agree with what you got by bruteforce counting.
Show that during the quasistatic isothermal expansion of a monatomic ideal gas, the change in entropy is related to the heat input by the simple formula
In the following chapter I'll prove that this formula is valid for any quasistatic process. Show, however, that it is not valid for the free expansion process described above.
What do you think about this solution?
We value your feedback to improve our textbook solutions.