Chapter 2: Q. 2.11 (page 60)
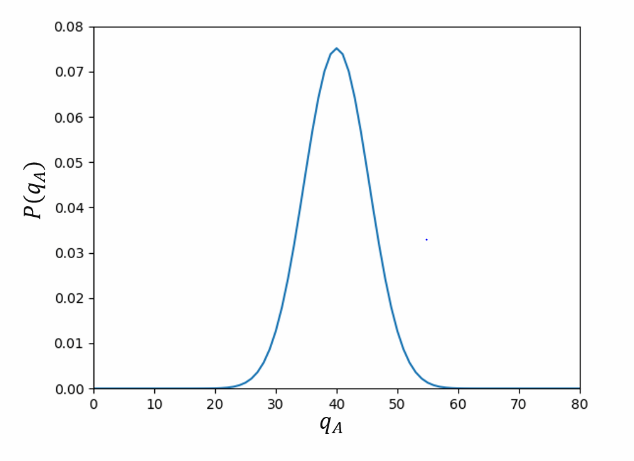
Use a computer to produce a table and graph, like those in this section, for two interacting two-state paramagnets, each containing elementary magnetic dipoles. Take a "unit" of energy to be the amount needed to flip a single dipole from the "up" state (parallel to the external field) to the "down" state (antiparallel). Suppose that the total number of units of energy, relative to the state with all dipoles pointing up, is; this energy can be shared in any way between the two paramagnets. What is the most probable macrostate, and what is its probability? What is the least probable macrostate, and what is its probability?
Short Answer
The most likely macrostate is when the energy units are evenly distributed, , with a probability of . The least likely state is when all the energy units are in partition or , or when , with a chance of .