Chapter 7: Q. 7.2 (page 260)
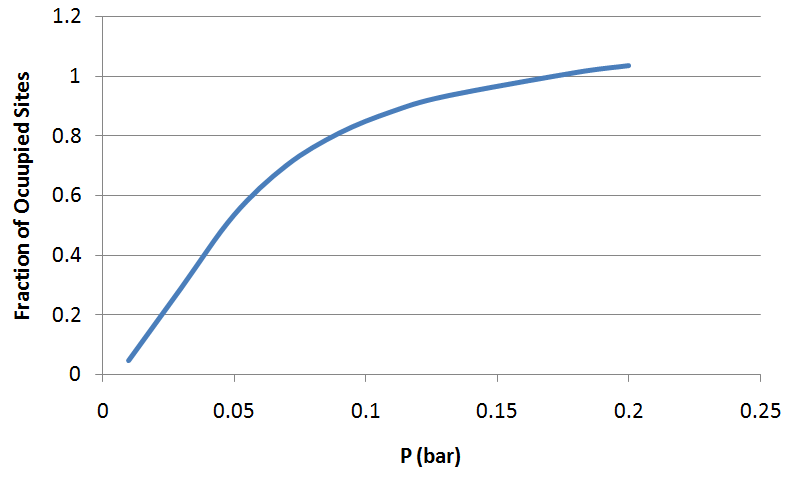
In a real hemoglobin molecule, the tendency of oxygen to bind to a heme site increases as the other three heme sites become occupied. To model this effect in a simple way, imagine that a hemoglobin molecule has just two sites, either or both of which can be occupied. This system has four possible states (with only oxygen present). Take the energy of the unoccupied state to be zero, the energies of the two singly occupied states to be , and the energy of the doubly occupied state to be (so the change in energy upon binding the second oxygen is ). As in the previous problem, calculate and plot the fraction of occupied sites as a function of the effective partial pressure of oxygen. Compare to the graph from the previous problem (for independent sites). Can you think of why this behavior is preferable for the function of hemoglobin?
Short Answer
Occupancy rises gradually for smaller values of . This shows that with cooperative binding, hemoglobin reacts with oxygen much fast in cells when is small.