Chapter 7: Q. 7.66 (page 323)
Short Answer
bffdb
Learning Materials
Features
Discover
Chapter 7: Q. 7.66 (page 323)
bffdb
All the tools & learning materials you need for study success - in one app.
Get started for free
The argument given above for why does not depend on the details of the energy levels available to the fermions, so it should also apply to the model considered in Problem 7.16: a gas of fermions trapped in such a way that the energy levels are evenly spaced and non-degenerate.
(a) Show that, in this model, the number of possible system states for a given value of q is equal to the number of distinct ways of writing q as a sum of positive integers. (For example, there are three system states for q = 3, corresponding to the sums 3, 2 + 1, and 1 + 1 + 1. Note that 2 + 1 and 1 + 2 are not counted separately.) This combinatorial function is called the number of unrestricted partitions of q, denoted p(q). For example, p(3) = 3.
(b) By enumerating the partitions explicitly, compute p(7) and p(8).
(c) Make a table of p(q) for values of q up to 100, by either looking up the values in a mathematical reference book, or using a software package that can compute them, or writing your own program to compute them. From this table, compute the entropy, temperature, and heat capacity of this system, using the same methods as in Section 3.3. Plot the heat capacity as a function of temperature, and note that it is approximately linear.
(d) Ramanujan and Hardy (two famous mathematicians) have shown that when q is large, the number of unrestricted partitions of q is given approximately by
Check the accuracy of this formula for q = 10 and for q = 100. Working in this approximation, calculate the entropy, temperature, and heat capacity of this system. Express the heat. capacity as a series in decreasing powers of , assuming that this ratio is large and keeping the two largest terms. Compare to the numerical results you obtained in part (c). Why is the heat capacity of this system independent of N, unlike that of the three dimensional box of fermions discussed in the text?
Evaluate the integral in equation numerically, to confirm the value quoted in the text.
Consider a system consisting of a single impurity atom/ion in a semiconductor. Suppose that the impurity atom has one "extra" electron compared to the neighboring atoms, as would a phosphorus atom occupying a lattice site in a silicon crystal. The extra electron is then easily removed, leaving behind a positively charged ion. The ionized electron is called a conduction electron, because it is free to move through the material; the impurity atom is called a donor, because it can "donate" a conduction electron. This system is analogous to the hydrogen atom considered in the previous two problems except that the ionization energy is much less, mainly due to the screening of the ionic charge by the dielectric behavior of the medium.
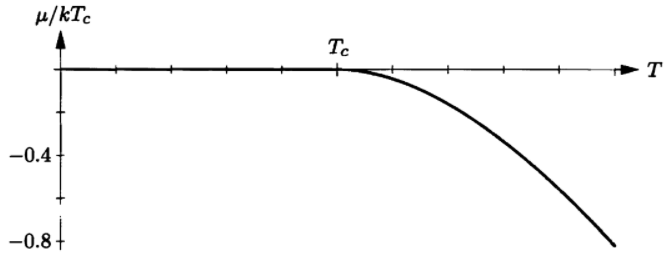
If you have a computer system that can do numerical integrals, it's not particularly difficult to evaluate for .
(a) As usual when solving a problem on a computer, it's best to start by putting everything in terms dimensionless variables. So define . Express the integral that defines , equation , in terms of these variables, you should obtain the equation
(b) According to given figure , the correct value of when , is approximately . Plug in these values and check that the equation above is approximately satisfied.
(c) Now vary , holding fixed, to find the precise value of for . Repeat for values ofranging from up to , in increments of . Plot a graph of as a function of temperature.
Consider the electromagnetic radiation inside a kiln, with a volume of V= I m3 and a temperature of 1500 K.
(a) What is the total energy of this radiation?
(b) Sketch the spectrum of the radiation as a function of photon energy.
(c) What fraction of all the energy is in the visible portion of the spectrum, with wavelengths between 400 nm and 700 nm?
What do you think about this solution?
We value your feedback to improve our textbook solutions.