Chapter 7: 7.32 (page 285)
Although the integrals (and ) forand cannot be
carried out analytically for all T, it's not difficult to evaluate them numerically
using a computer. This calculation has little relevance for electrons in metals (for
which the limit is always sufficient), but it is needed for liquid and
for astrophysical systems like the electrons at the center of the sun.
(a) As a warm-up exercise, evaluate theintegral () for the case
and , and check that your answer is consistent with the graph shown
above. (Hint: As always when solving a problem on a computer, it's best to
first put everything in terms of dimensionless variables. So let role="math" localid="1649996205331"
, and . Rewrite everything in terms of these variables,
and then put it on the computer.)
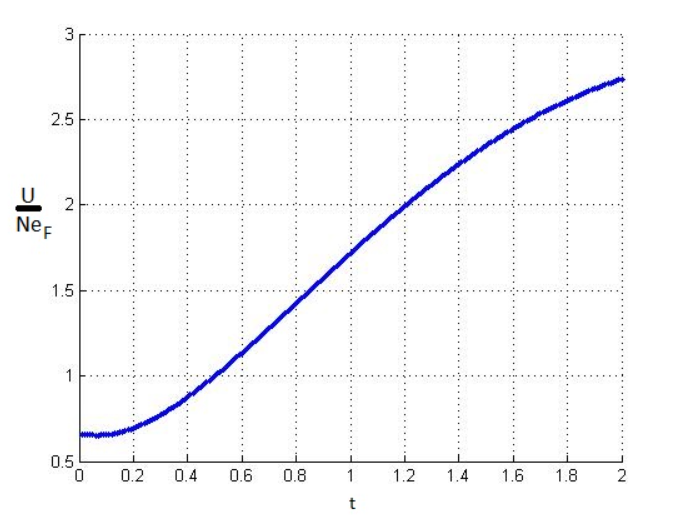
(b) The next step is to vary holding fixed, until the integral works out to
the desired value,. Do this for values of ranging from up to ,
and plot the results to reproduce Figure. (It's probably not a good idea
to try to use numerical methods when is much smaller than , since
you can start getting overflow errors from exponentiating large numbers.
But this is the region where we've already solved the problem analytically.)
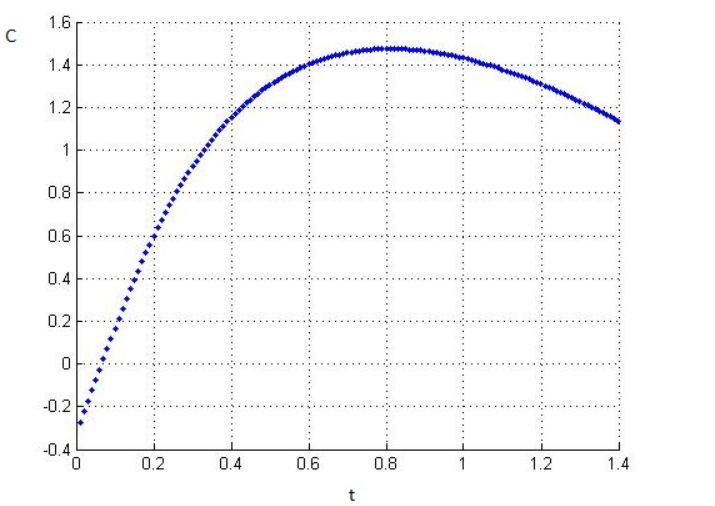
(c) Plug your calculated values ofµ into the energy integral (), and evaluate
that integral numerically to obtain the energy as a function of temperature
forup to Plot the results, and evaluate the slope to obtain the
heat capacity. Check that the heat capacity has the expected behavior at
both low and high temperatures.
Short Answer
a) The value of integral is .
b) The value of
c) We get the final equation as
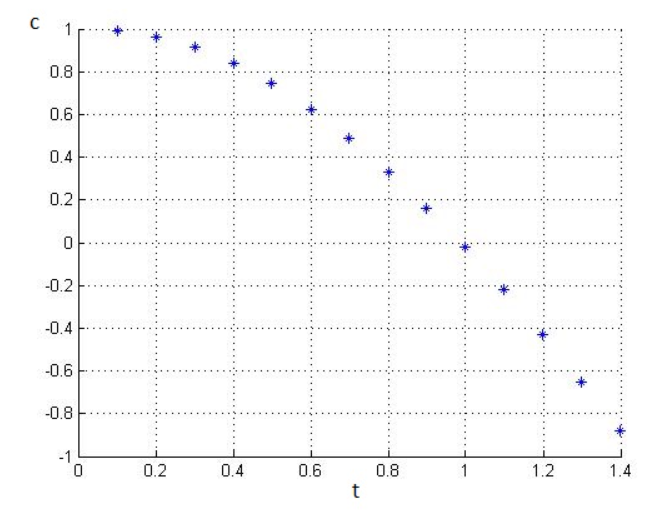
 Here, the equation between and from graph can be seen ,
Here, the equation between and from graph can be seen ,