Chapter 3: Q. 3.17 (page 103)
Verify every entry in the third line of Table 3.2 (starting with .
Short Answer
All the entries in the third line are verified.
Learning Materials
Features
Discover
Chapter 3: Q. 3.17 (page 103)
Verify every entry in the third line of Table 3.2 (starting with .
All the entries in the third line are verified.
All the tools & learning materials you need for study success - in one app.
Get started for free
Use a computer to study the entropy, temperature, and heat capacity of an Einstein solid, as follows. Let the solid contain 50 oscillators (initially), and from 0 to 100 units of energy. Make a table, analogous to Table 3.2, in which each row represents a different value for the energy. Use separate columns for the energy, multiplicity, entropy, temperature, and heat capacity. To calculate the temperature, evaluate for two nearby rows in the table. (Recall that for some constant .) The heat capacity can be computed in a similar way. The first few rows of the table should look something like this:
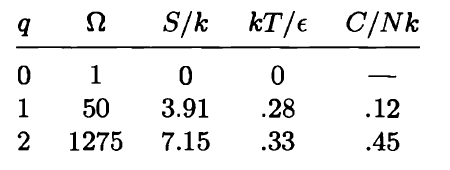
(In this table I have computed derivatives using a "centered-difference" approximation. For example, the temperature is computed as .) Make a graph of entropy vs. energy and a graph of heat capacity vs. temperature. Then change the number of oscillators to 5000 (to "dilute" the system and look at lower temperatures), and again make a graph of heat capacity vs. temperature. Discuss your prediction for the heat capacity, and compare it to the data for lead, aluminum, and diamond shown in Figure 1.14. Estimate the numerical value of in electron-volts, for each of those real solids.
Show that the entropy of a two-state paramagnet, expressed as a function of temperature, is , where . Check that this formula has the expected behavior as and .
Use the result of Problem 2.42 to calculate the temperature of a black hole, in terms of its mass . (The energy is . ) Evaluate the resulting expression for a one-solar-mass black hole. Also sketch the entropy as a function of energy, and discuss the implications of the shape of the graph.
An ice cube (mass )is left sitting on the kitchen table, where it gradually melts. The temperature in the kitchen is .
(a) Calculate the change in the entropy of the ice cube as it melts into water at . (Don't worry about the fact that the volume changes somewhat.)
(b) Calculate the change in the entropy of the water (from the melted ice) as its temperature rises from to .
(c) Calculate the change in the entropy of the kitchen as it gives up heat to the melting ice/water.
(d) Calculate the net change in the entropy of the universe during this process. Is the net change positive, negative, or zero? Is this what you would expect?
In order to take a nice warm bath, you mix 50 liters of hot water at with 25 liters of cold water at . How much new entropy have you created by mixing the water?
What do you think about this solution?
We value your feedback to improve our textbook solutions.