Chapter 3: Q. 3.24 (page 107)
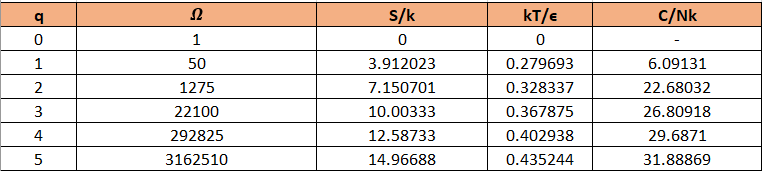
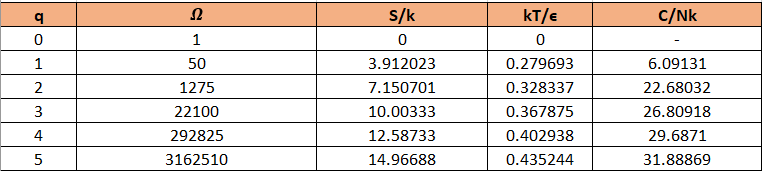
Use a computer to study the entropy, temperature, and heat capacity of an Einstein solid, as follows. Let the solid contain 50 oscillators (initially), and from 0 to 100 units of energy. Make a table, analogous to Table 3.2, in which each row represents a different value for the energy. Use separate columns for the energy, multiplicity, entropy, temperature, and heat capacity. To calculate the temperature, evaluate for two nearby rows in the table. (Recall that for some constant .) The heat capacity can be computed in a similar way. The first few rows of the table should look something like this:
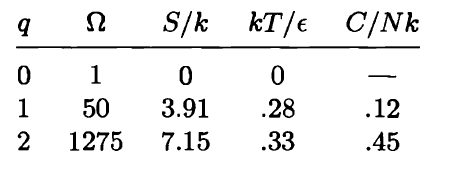
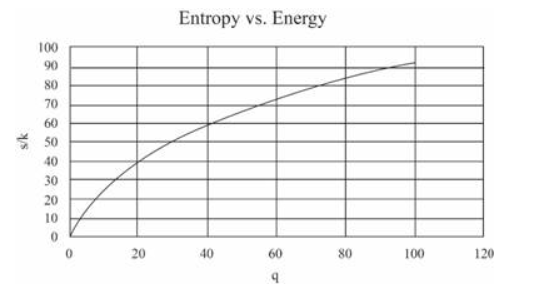
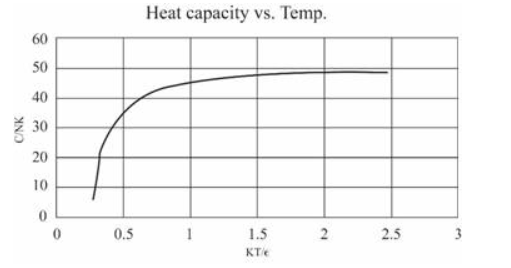
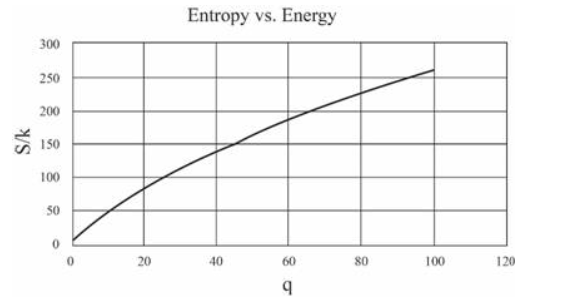
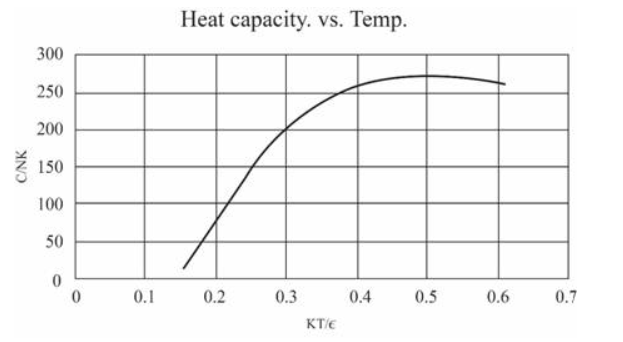
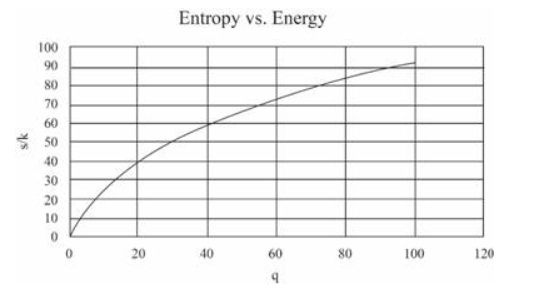
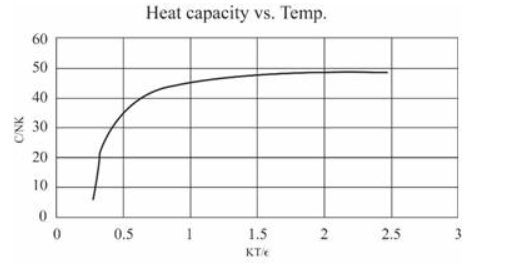
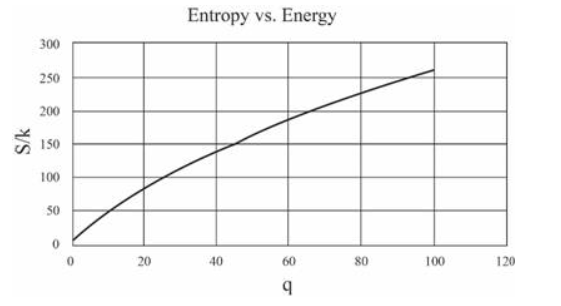
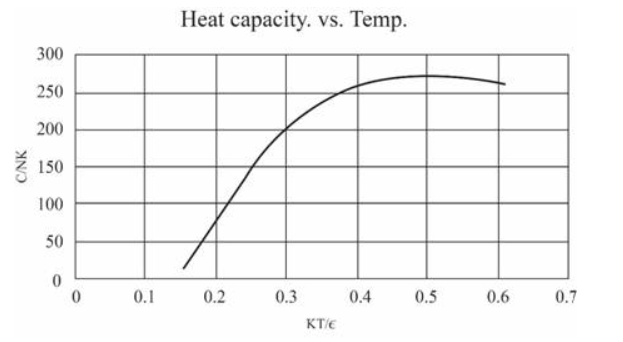
(In this table I have computed derivatives using a "centered-difference" approximation. For example, the temperature is computed as .) Make a graph of entropy vs. energy and a graph of heat capacity vs. temperature. Then change the number of oscillators to 5000 (to "dilute" the system and look at lower temperatures), and again make a graph of heat capacity vs. temperature. Discuss your prediction for the heat capacity, and compare it to the data for lead, aluminum, and diamond shown in Figure 1.14. Estimate the numerical value of in electron-volts, for each of those real solids.
Short Answer
The table can be prepared as:
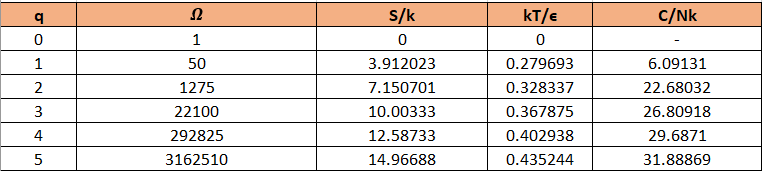
The graphs can be made as:
For 50 oscillations
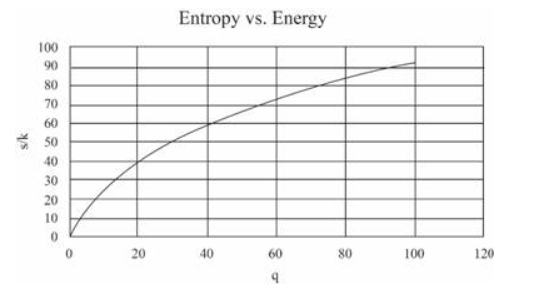
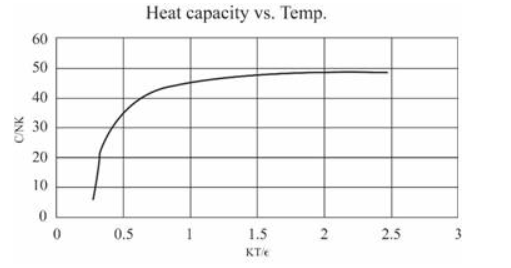
For 5000 oscillations
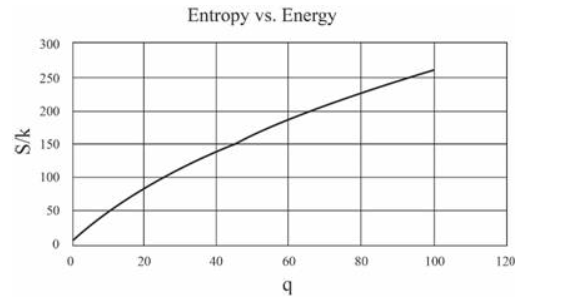
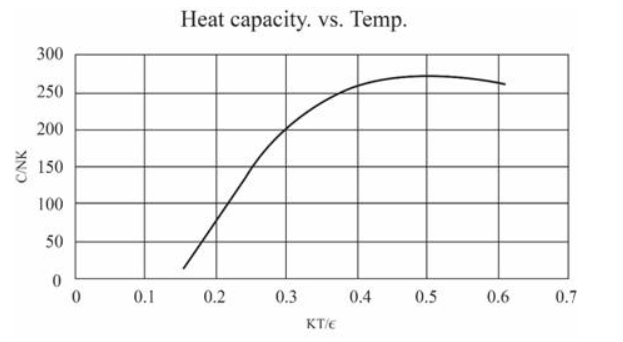
The values of are: