Chapter 3: Q. 3.7 (page 92)
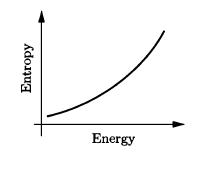
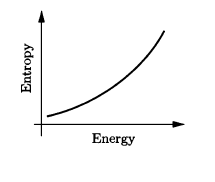
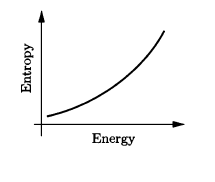
Use the result of Problem 2.42 to calculate the temperature of a black hole, in terms of its mass . (The energy is . ) Evaluate the resulting expression for a one-solar-mass black hole. Also sketch the entropy as a function of energy, and discuss the implications of the shape of the graph.
Short Answer
The required expression is and for one-solar-mass of a black hole, the temperature can be calculated be .
The graph below depicts the entropy as a function of energy which is a concave up graph.