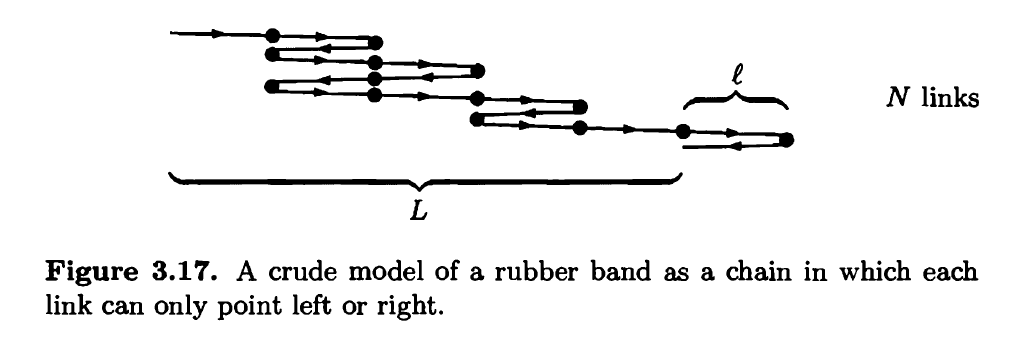
Polymers, like rubber, are made of very long molecules, usually tangled up in a configuration that has lots of entropy. As a very crude model of a rubber band, consider a chain of N links, each of length (see Figure 3.17). Imagine that each link has only two possible states, pointing either left or right. The total length L of the rubber band is the net displacement from the beginning of the first link to the end of the last link.
(a) Find an expression for the entropy of this system in terms of N and NR, the number of links pointing to the right.
(b) Write down a formula for L in terms of N and NR.
(c) For a one-dimensional system such as this, the length L is analogous to the volume V of a three-dimensional system. Similarly, the pressure P is replaced by the tension force F. Taking F to be positive when the rubber band is pulling inward, write down and explain the appropriate thermodynamic identity for this system.
(d) Using the thermodynamic identity, you can now express the tension force F in terms of a partial derivative of the entropy. From this expression, compute the tension in terms of L, T, N, and .
(e) Show that when L << N, the tension force is directly proportional to L (Hooke's law).
(f) Discuss the dependence of the tension force on temperature. If you increase the temperature of a rubber band, does it tend to expand or contract? Does this behavior make sense?
(g) Suppose that you hold a relaxed rubber band in both hands and suddenly stretch it. Would you expect its temperature to increase or decrease? Explain. Test your prediction with a real rubber band (preferably a fairly heavy one with lots of stretch), using your lips or forehead as a thermometer. (Hint: The entropy you computed in part (a) is not the total entropy of the rubber band. There is additional entropy associated with the vibrational energy of the molecules; this entropy depends on U but is approximately independent of L.)





