Chapter 5: Q 5.37 (page 176)
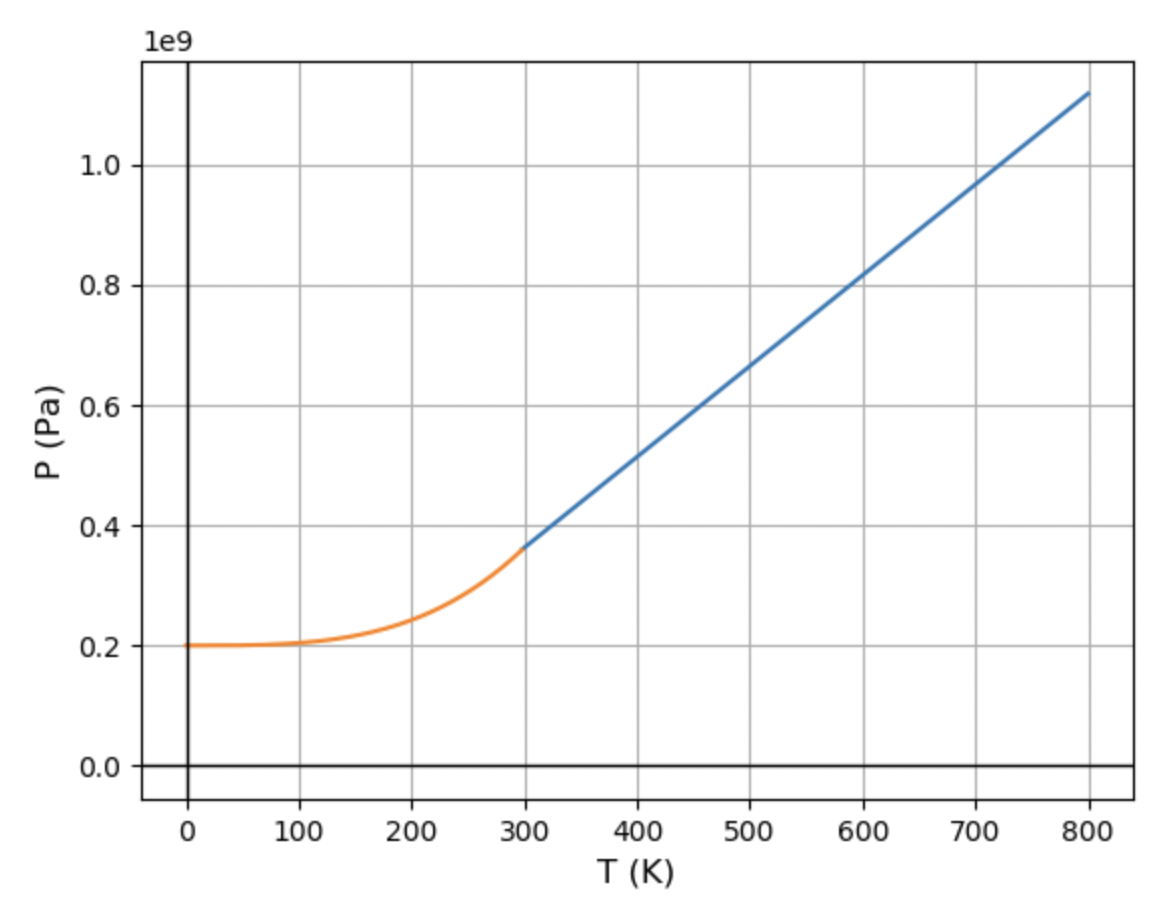
Use the data at the back of this book to calculate the slope of the calcite-aragonite phase boundary (at 298 K). You located one point on this phase boundary in Problem 5.28; use this information to sketch the phase diagram of calcium carbonate.
Short Answer
The calcium carbonate phase diagram would be linear up to temperature 58.2 SK, but as the temperature decreased, the pressure would not fall linearly, resulting in a non linear graph