Chapter 5: Q 5.62 (page 194)
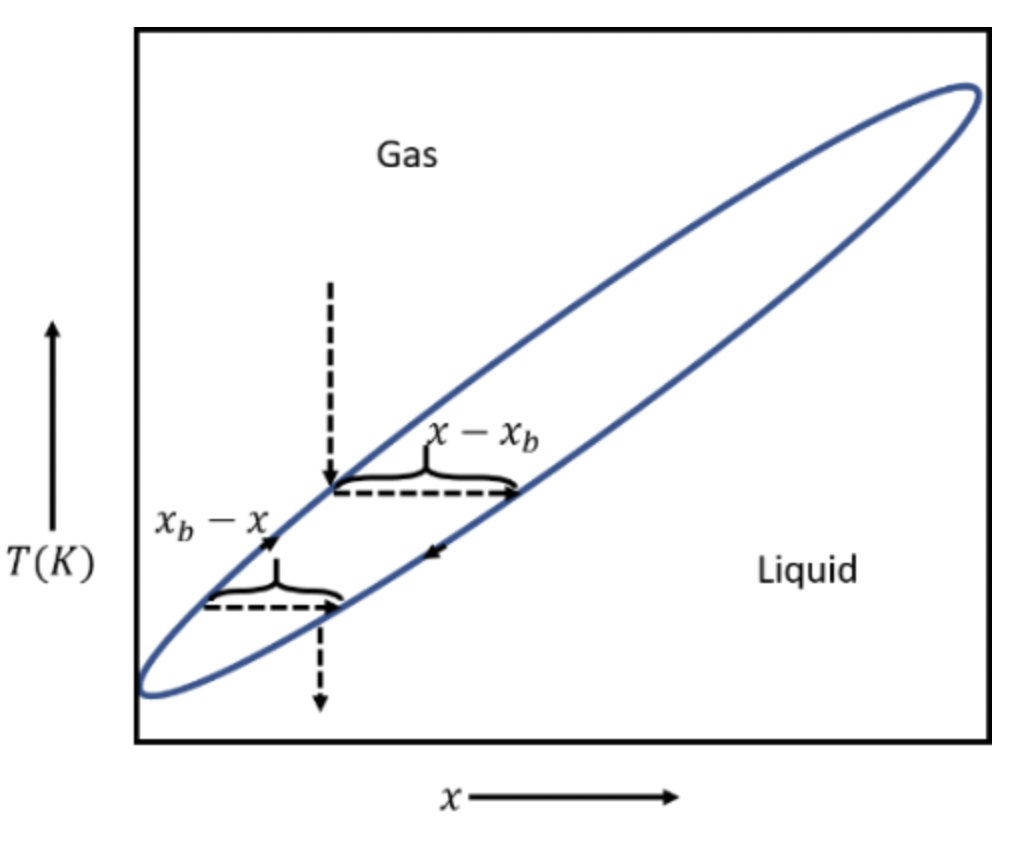
Consider a completely miscible two-component system whose overall composition is x, at a temperature where liquid and gas phases coexist. The composition of the gas phase at this temperature is and the composition of the liquid phase is . Prove the lever rule, which says that the proportion of liquid to gas is . Interpret this rule graphically on a phase diagram.
Short Answer
Hence proved