Chapter 5: Q.5.36 (page 175)
Effect of altitude on boiling water.
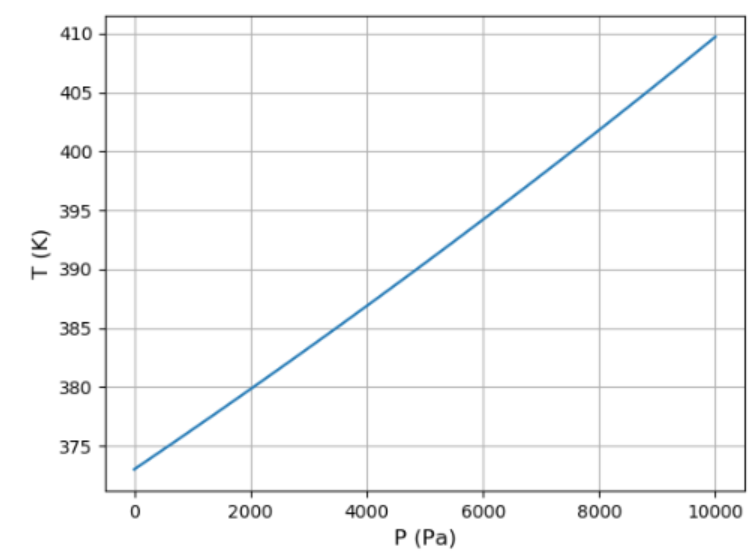
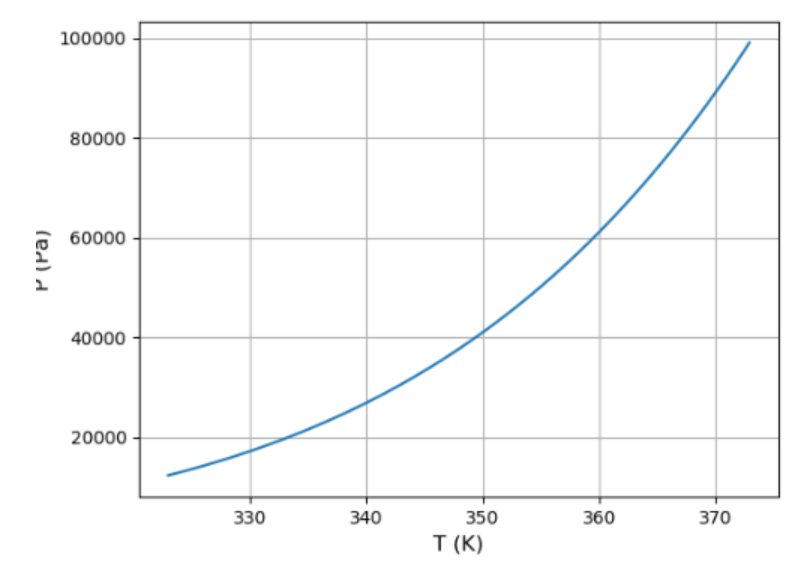
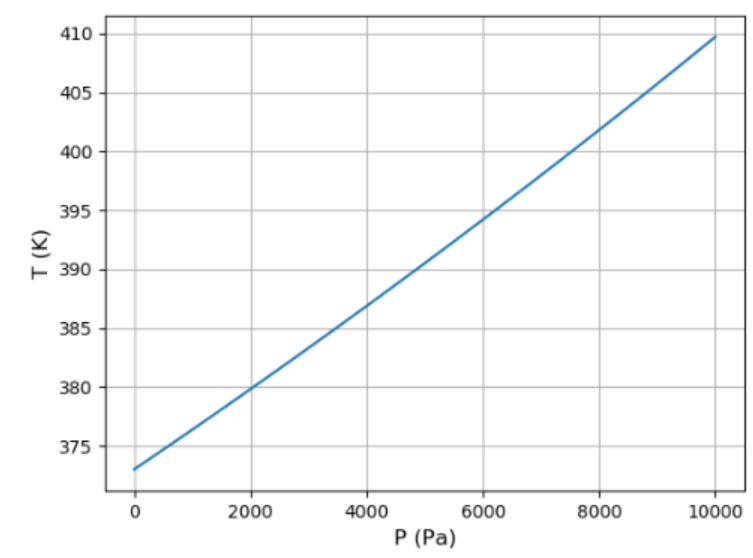
(a) Use the result of the previous problem and the data in Figure 5.11 to plot a graph of the vapor pressure of water between and . How well can you match the data at the two endpoints?
(b) Reading the graph backward, estimate the boiling temperature of water at each of the locations for which you determined the pressure in Problem 1.16. Explain why it takes longer to cook noodles when you're camping in the mountains.
(c) Show that the dependence of boiling temperature on altitude is very nearly (though not exactly) a linear function, and calculate the slope in degrees Celsius per thousand feet (or in degrees Celsius per kilometer).
Short Answer
a). The variation of vapour pressure of water between and is shown.
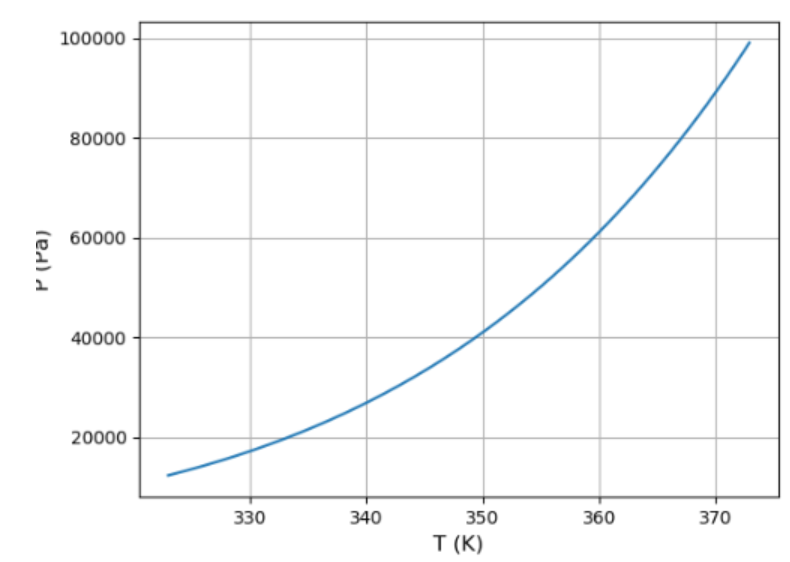
b). The estimate values of the boiling temperature of water for different altitudes is .
c). A graph to show the variation of the altitude with pressure.