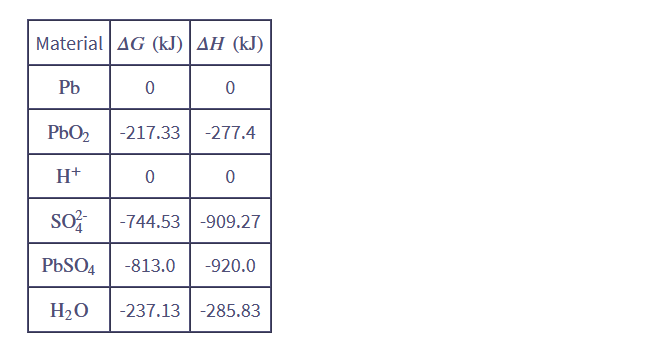
Chapter 5: Q. 5.3 (page 155)
Use the data at the back of this book to verify the values of ΔH and ΔGquoted above for the lead-acid reaction 5.13.
At temp 298K and pressure 1 bar.
Short Answer
The value of Gibbs free energy = -315.72 kJ.
Learning Materials
Features
Discover
Chapter 5: Q. 5.3 (page 155)
Use the data at the back of this book to verify the values of ΔH and ΔGquoted above for the lead-acid reaction 5.13.
At temp 298K and pressure 1 bar.

The value of Gibbs free energy = -315.72 kJ.
All the tools & learning materials you need for study success - in one app.
Get started for free
The standard enthalpy change upon dissolving one mole of oxygen at 25°C is -11.7 kJ. Use this number and the van't Hoff equation (Problem 5.85) to calculate the equilibrium (Henry's law) constant for oxygen in water at 0°C and at 100° C. Discuss the results briefly.
Suppose you have a liquid (say, water) in equilibrium with its gas phase, inside some closed container. You then pump in an inert gas (say, air), thus raising the pressure exerted on the liquid. What happens?
(a) For the liquid to remain in diffusive equilibrium with its gas phase, the chemical potentials of each must change by the same amount: Use this fact and equation 5.40 to derive a differential equation for the equilibrium vapour pressure, Pv as a function of the total pressure P. (Treat the gases as ideal, and assume that none of the inert gas dissolves in the liquid.)
(b) Solve the differential equation to obtain
where the ratio V/N in the exponent is that of the liquid. (The term Pv(Pv) is just the vapour pressure in the absence of the inert gas.) Thus, the presence of the inert gas leads to a slight increase in the vapour pressure: It causes more of the liquid to evaporate.
(c) Calculate the percent increase in vapour pressure when air at atmospheric pressure is added to a system of water and water vapour in equilibrium at 25°C. Argue more generally that the increase in vapour pressure due to the presence of an inert gas will be negligible except under extreme conditions.
A muscle can be thought of as a fuel cell, producing work from the metabolism of glucose:
(a) Use the data at the back of this book to determine the values of and for this reaction, for one mole of glucose. Assume that the reaction takes place at room temperature and atmospheric pressure.
(b) What is maximum amount of work that a muscle can perform , for each mole of glucose consumed, assuming ideal operation?
(c) Still assuming ideal operation, how much heat is absorbed or expelled by the chemicals during the metabolism of a mole of glucose?
(d) Use the concept of entropy to explain why the heat flows in the direction it does?
(e) How would your answers to parts (a) and (b) change, if the operation of the muscle is not ideal?
Use a Maxwell relation from the previous problem and the third law of thermodynamics to prove that the thermal expansion coefficient (defined in Problem 1.7) must be zero at T=0.
Consider a completely miscible two-component system whose overall composition is x, at a temperature where liquid and gas phases coexist. The composition of the gas phase at this temperature is and the composition of the liquid phase is . Prove the lever rule, which says that the proportion of liquid to gas is . Interpret this rule graphically on a phase diagram.
What do you think about this solution?
We value your feedback to improve our textbook solutions.