Chapter 0: Q44P (page 1)
Write thereaction for trichloroacetic acid,,for anilinium ion,  , and for lanthanum ion,.
, and for lanthanum ion,.
Short Answer
Thereaction for all is given in the solution.
Learning Materials
Features
Discover
Chapter 0: Q44P (page 1)
Write thereaction for trichloroacetic acid,,for anilinium ion,  , and for lanthanum ion,.
, and for lanthanum ion,.
Thereaction for all is given in the solution.
All the tools & learning materials you need for study success - in one app.
Get started for free
When you flip a coin, the probability of its landing on each side isin Equations 28-2 and 28-3. If you flip it times, the expected number of heads equals the expected number of tailsThe expected standard deviation for flips is. From Table 4-1, we expect thatof the results will lie withinand of the results will lie within.
(a) Find the expected standard deviation for the number of heads incoin flips.
(b) By interpolation in Table 4-1, find the value of that includesof the area of the Gaussian curve. We expect thatof the results will lie within this number of standard deviations from the mean.
(c) If you repeat thecoin flips many times, what is the expected range for the number of heads that includesof the results? (For example, your answer might be, "The rangetowill be observedof the time.")
Control chart -- Volatile compounds in human blood serum were measured by purge and trap gas chromatography/mass spectrometry. For quality control, serum was periodically spiked with a constant amount of -dichlorobenzene and the concentration role="math" localid="1663334314380" was measured. Find the mean and standard deviation for the following spike data and prepare a control chart. State whether or not the observations (Obs.) meet each criterion for stability in a control chart.
23.For a 0.1Maqueous solution of sodium acetate,, one mass alance is simply. Write a mass balance involving acetate.
Protein and carbohydrates provide, whereas fat gives . (Remember that 1 Calorie , with a capital C, is really .) The weight percentages of these components in some foods are
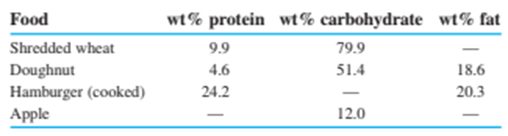
Calculate the number of calories per gram and calories per ounce in each of these foods. (Use Table 1-4to convert grams into ounces, remembering that there are 16ounces in 1pound.)
List the steps in a chemical analysis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.