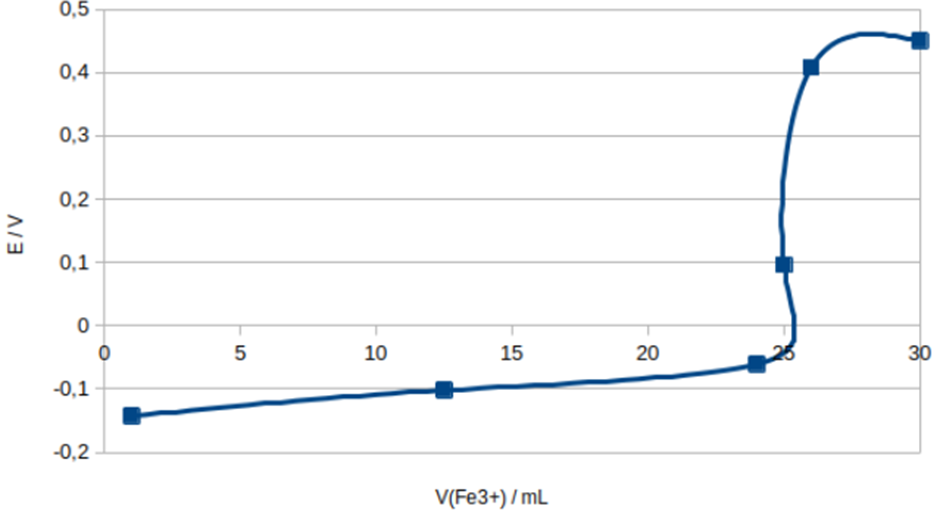
d)
Calculate the equivalency point using data from the job before proceeding with potential calculations for each extra volume:
Now determine three titration regions:
1. Before the equivalence point -1.00,12.50 and 24.0mL .
2. At the equivalence point -25.0mL .
3. After the equivalence point -26.0 and 30.0mL .
There is an excess of in the solution at 1.00 mL of added before equivalence point, so use equation to compute the potential by simply replacing concentrations of ions with their volume of excess is 24/25 .
Now use equation to calculate potential:
The volume of is one-half of the quantity required for the equivalence point at 12.5 mL , therefore the log term is and the potential equation is:
For the task that means:
role="math" localid="1664871633983"
Apply the same calculation used at , substituting the volume of produced and surplus iron:
At 25 mL, the equivalence point, and are at equilibrium and also so write:
Now calculate the potential by adding the equations together:
role="math" localid="1664872109070"
By rearranging the equation,
By inserting the concentrations of cerium,
Now calculate :
The total potential at 25 mL is:
There is an excess of in the solution after equivalence point at 25 mL of additional , therefore use equation (2) to determine the potential by simply replacing concentrations of Tl ions with their corresponding volume.
For example, if 26mL of was added, the volume of formed is 25/25 and the volume of excess is 1/25.
Now use equation (2) to calculate potential:
Now substitute the volume of extra for 30mL of additional:
Now sketch the titration curve with added volume of on x-axis and potential on y-axis: