Chapter 10: QBE (page 229)
(c) How many milliliters of should be added to of to give a pH of in ?
Short Answer
The volume of nitric acid is = 61.6mL
Learning Materials
Features
Discover
Chapter 10: QBE (page 229)
(c) How many milliliters of should be added to of to give a pH of in ?
The volume of nitric acid is = 61.6mL
All the tools & learning materials you need for study success - in one app.
Get started for free
A dibasic compound, B, has pKb1 5 4.00 and pKb2 5 6.00.
Find the fraction in the form at pH 7.00, using Equation
10-19. Note that K1 and K2 in Equation 10-19 are acid dissociation
constants forlocalid="1655450143786"
1 (K1 = Kw/Kb2 and K2 = Kw/Kb1).
Write the general structure of an amino acid. Why do some amino acids in Tablehave two pK values and others three?
Calculate for cis-butenedioic acid at pH 1.00, 1.92, 6.00, 6.27 and 10.00.
Fractional composition in a triprotic system. For a triprotic system, the fractional composition equations where areUse these equations to create a fractional composition diagram analogous to Figure 10-4 for the amino acid tyrosine. What is the fraction of each species at?
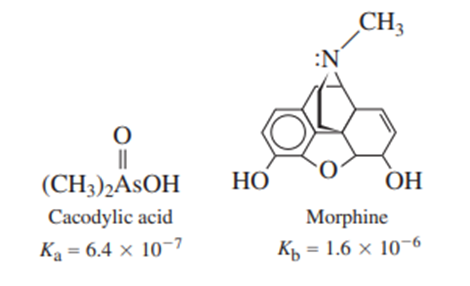
A solution was prepared from 10.0 mLof 0.100Mcacodylic acid and 10.0mLof 0.0800MNaOH. To this mixture was added 1.00mL of morphine. Calling morphine B, calculate the fraction of morphine present in the form .
What do you think about this solution?
We value your feedback to improve our textbook solutions.