Chapter 10: Q33P (page 232)
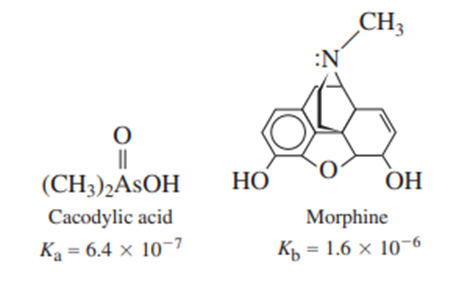
A solution was prepared from 10.0 mLof 0.100Mcacodylic acid and 10.0mLof 0.0800MNaOH. To this mixture was added 1.00mL of morphine. Calling morphine B, calculate the fraction of morphine present in the form .
Short Answer
The fraction of morphine in the form is 0.96

