Chapter 10: Q32P (page 232)
A solution containing acetic acid, oxalic acid, ammonia, and pyridine has a of 9.00. What fraction of ammonia is not protonated?
Short Answer
The fraction of unprotonated ammonia for the given solution is .
Learning Materials
Features
Discover
Chapter 10: Q32P (page 232)
A solution containing acetic acid, oxalic acid, ammonia, and pyridine has a of 9.00. What fraction of ammonia is not protonated?
The fraction of unprotonated ammonia for the given solution is .
All the tools & learning materials you need for study success - in one app.
Get started for free
The base B has = 5.00.
(a) What is the value of for the acid ?
(b) At what pH is [] = [B]?
(c) Which is the principal species at pH 7.00: B or ?
(d) What is the quotient [B] / [] at pH 12.00?
A solution was prepared from 10.0 mLof 0.100Mcacodylic acid and 10.0mLof 0.0800MNaOH. To this mixture was added 1.00mL of morphine. Calling morphine B, calculate the fraction of morphine present in the form .
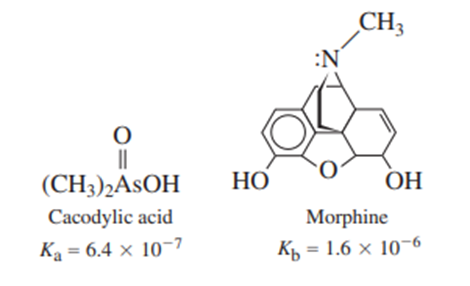
Find the pH of with Equation.
localid="1654853037629"
(Yes, the values oflocalid="1654853051658" andlocalid="1654853058271" are equal.)
(a) Use these equilibrium constants to prepare a fractional composition diagram for this tetraprotic system.
(b) You should do this part with your head and your calculator, not your spreadsheet. The solubility of is given by
localid="1654853063951"
What concentration of localid="1654853075036" is in equilibrium with solid localid="1654853085944" ?
(c) What concentration oflocalid="1654853094735" is in equilibrium with localid="1654853101499" if the solution localid="1654853109266" is adjusted tolocalid="1654853117749" ?
The diprotic acid has and .
(a) At what pH is = ?
(b) At what pH is = ?
(c) Which is the principal species at pH 2.00: or ?
(d) Which is the principal species at pH 6.00?
(e) Which is the principal species at pH 10.00?
What do you think about this solution?
We value your feedback to improve our textbook solutions.