Chapter 10: Q10FE (page 230)
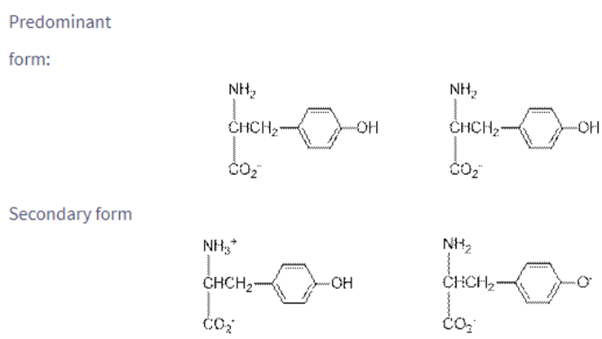
Draw the structures of the predominant forms of glutamic acid and tyrosine at and What is the second most abundant species at each ?.
Short Answer
The Predominant form, Secondary form of given pH drawn.
Learning Materials
Features
Discover
Chapter 10: Q10FE (page 230)
Draw the structures of the predominant forms of glutamic acid and tyrosine at and What is the second most abundant species at each ?.
The Predominant form, Secondary form of given pH drawn.

All the tools & learning materials you need for study success - in one app.
Get started for free
(c) How many milliliters of should be added to of to give a pH of in ?
In this problem, we calculate the pH of the intermediate form of a diprotic acid, taking activities into account.
(a) Including activity coefficients, derive Equation 10 - 11 for potassium hydrogen phthalate in the example following Equation 10 - 12 ).
(b) Calculate the pH of , using the results in part (a). Assume that the sizes of both and are 600pm . For comparison, Equation 10 - 11 gives .
Consider the diprotic acidwithand.Find the pH and concentrations of, andin
(a)role="math" localid="1654926233413"
(b) role="math" localid="1654926403235" ;
(c) .
Neutral lysine can be written HL. The other forms of lysine areand. The isoelectric point is the pH at which the average charge of lysine is . Therefore, at the isoelectric point,. Use this condition to calculate the isoelectric pH of lysine.
How many millilitres of 1.00 M KOH should be added to 100 mL of solution containing 10.0 g of histidine hydrochloride
[His? HCI (HisH1) (CI2), FM 191.62 ] to get a pH of 9.30 ?
What do you think about this solution?
We value your feedback to improve our textbook solutions.