Chapter 10: Q21P (page 231)
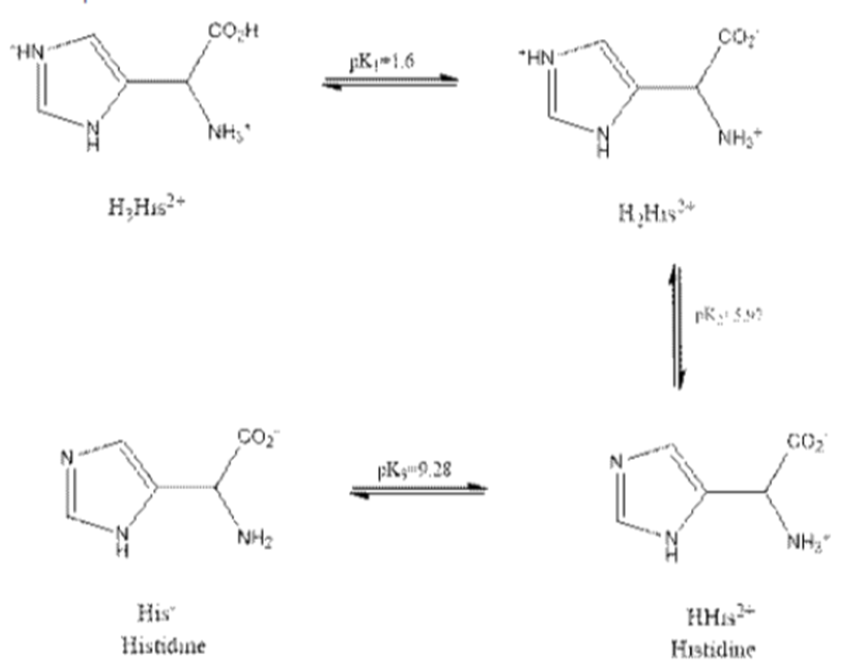
How many millilitres of 1.00 M KOH should be added to 100 mL of solution containing 10.0 g of histidine hydrochloride
[His? HCI (HisH1) (CI2), FM 191.62 ] to get a pH of 9.30 ?
Short Answer
The required amount of Potassium hydroxide solution 78.9mL