Chapter 22: Q3TY (page 574)
Would the answers be different if m/z 5 500 were an ion with a mass
of 1 000 Da and z 5 2?
Short Answer
No the answer will be same.
Learning Materials
Features
Discover
Chapter 22: Q3TY (page 574)
Would the answers be different if m/z 5 500 were an ion with a mass
of 1 000 Da and z 5 2?
No the answer will be same.
All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Find the number of rings plus double bonds in a molecule with the composition C14H12and draw one plausible structure.
(b)For an ion or radical, the rings + double bonds formula gives noninteger answers because the formula is based on valences in neutral molecules with all electrons paired. How many rings plus double bonds are predicted for C4H10NO+? Draw one structure forC4H10NO+.
28: A limitation on how many spectra per second can be recorded by a time-of-flight mass spectrometer is the time it takes the slowest ions to go from the source to the detector. Suppose we want to scan up to m/z 500. Calculate the speed of this heaviest ion if it is accelerated through 5.00 kV in the source. How long would it take to drift 2.00 m through a spectrometer? At what frequency could you record spectra if a new extraction cycle were begun each time this heaviest ion reached the detector? What would be the frequency if you wanted to scan up to m/z 1000?
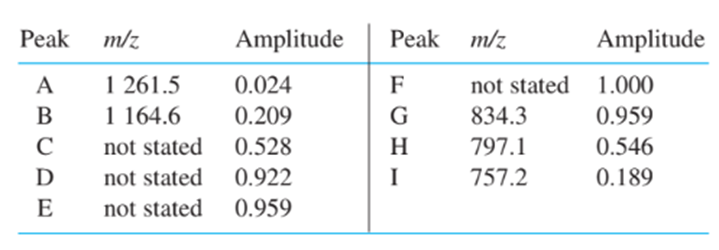
38: An electrospray–transmission quadrupole mass spectrum of the a chain of hemoglobin from acidic solution exhibits nine peaks corresponding to MHn1n . Find the charge, n, for peaks A–I. Calculate the molecular mass of the neutral protein, M, from peaks A, B, G, H, and I, and find the mean value.
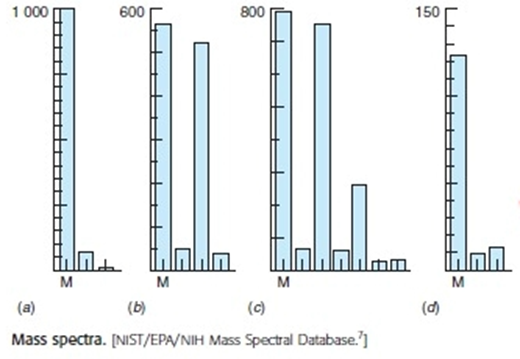
(This is a long exercise suitable for group work.) Relative intenities for the molecular ion region of several compounds are listed in arts (a)-(d) and shown in the figure. Suggest a composition for each nolecule and calculate the expected isotopic peak intensities.
a)
b)
c) m/z
d)
(a) Spectra A and B belong to isomers of C6H12O. Explain how you can tell which isomer goes with each spectrum.
What do you think about this solution?
We value your feedback to improve our textbook solutions.