Chapter 22: QEE (page 598)
(a) Spectra A and B belong to isomers of C6H12O. Explain how you can tell which isomer goes with each spectrum.
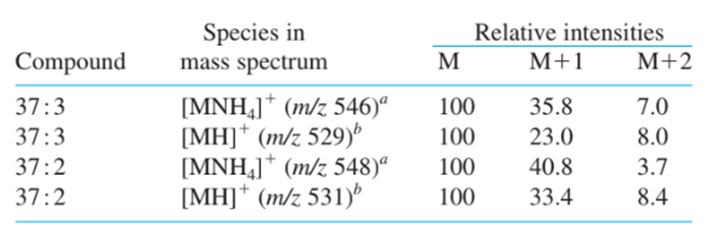
Short Answer
(a) The arrival of a strong peak at is the first difference between the two spectra. m/z = 72 It's possible that something in A isn't present in B.
(b) The intensity with which M + 1 in relation to M+ the following calculation can be used to find this. Intensity