Chapter 22: Q1IC (page 571)
Draw a rearrangement like reaction D in figure 22-12 to show how m/z 58 arises from 4-methyl-2-pentanone.
Short Answer
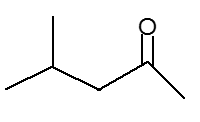
The structure of the 4-methyl-2-pentanone is shown below :
Learning Materials
Features
Discover
Chapter 22: Q1IC (page 571)
Draw a rearrangement like reaction D in figure 22-12 to show how m/z 58 arises from 4-methyl-2-pentanone.
The structure of the 4-methyl-2-pentanone is shown below :

All the tools & learning materials you need for study success - in one app.
Get started for free
34: What is collisionally activated dissociation? At what points in a mass spectrometer does it occur?
32:Why is direct electron ionization in Figure 22-14 permissible for gas chromatography, but only for capillary columns in liquidchromatography in Figure 22-29?
The molecular ion region in the mass spectrum of a large molecule, such as a protein, consists of a cluster of peaks differing by 1 Da. This pattern occurs because a molecule with many atoms has a high probability of containing one or several atoms of and . In fact, the probability of finding a molecule with only and may be so small that the nominal molecular ion is not observed. The electrospray mass spectrum of the rat protein interleukin-8 consists of a series of clusters of peaks arising from intact molecular ions with different charge. One cluster has peaks at m/z 1 961.12, 1 961.35, 1 961.63, 1 961.88, 1 962.12 (tallest peak), 1 962.36, 1 962.60, 1 962.87, 1 963.10, 1 963.34, 1 963.59, 1 963.85, and 1 964.09. These peaks correspond to isotopic ions differing by 1 Da. From the observed peak separation, fi nd the charge of the ions in this cluster. From m/z of the tallest peak, estimate the molecular mass of the protein.
Bone consists of the protein collagen and the mineral hydroxyapatite,. The content of archaeological humanskeletons measured by graphite furnace atomic absorption shedslight on customs and economic status of individuals in historicaltimes. 37 Explain why La3+is added to bone samples to suppressmatrix interference in Pbanalysis.
(a) Spectra A and B belong to isomers of C6H12O. Explain how you can tell which isomer goes with each spectrum.
What do you think about this solution?
We value your feedback to improve our textbook solutions.