Chapter 22: Q18 P (page 600)
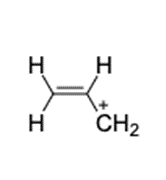
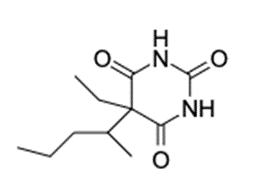
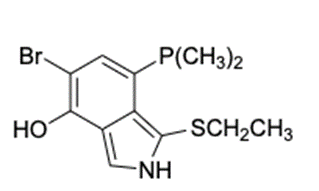
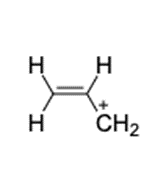
Find the number of rings 1 double bonds in molecules with the following compositions and draw one plausible structure for each: (a) (b)(c) fragment in a mass spectrum with the composition .
Short Answer
(a) The structure of is
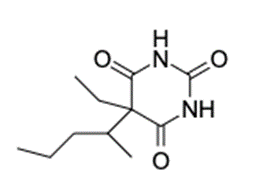
(b) The structure of is
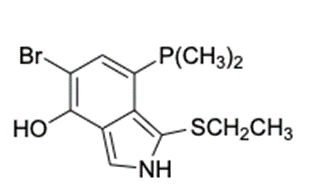
(c) The structure of is