Chapter 25: Q25P (page 711)
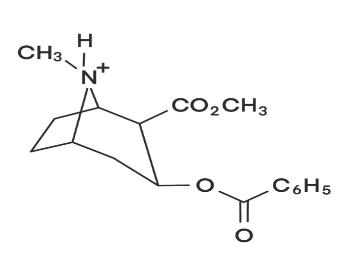
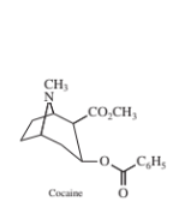
Chromatography–mass spectrometry. Cocaine metabolism in rats can be studied by injecting the drug and periodically with drawing blood to measure levels of metabolites by HPLC–mass spectrometry. For quantitative analysis, isotopically labelled internal standards are mixed with the blood sample. Blood was analysed by reversed-phase chromatography with an acidic eluent and atmospheric pressure chemical ionization mass spectrometry for detection. The mass spectrum of the collisionally activated dissociation products from the m/z 304 positive ion is shown in the figure on the next page. Selected reaction monitoring (m/z 304 from mass filter Q1 and m/z 182 from Q3 in Figure 22-33) gave a single chromatographic peak at 9.22 min for cocaine. The internal standard -cocaine gave a single peak at 9.19 min for m/z 309 (Q1) 182(Q3).
(a) Draw the structure of the ion at m/z 304.
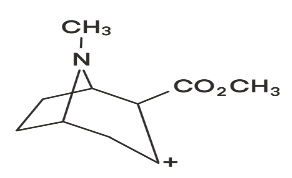
(b) Suggest a structure for the ion at m/z 182.
(c) The intense peaks at m/z 182 and 304 do not have isotopic partners at m/z 183 and 305. Explain why.
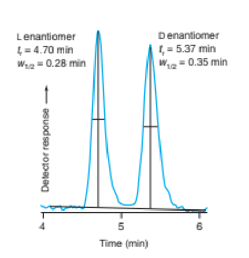
(d) Rat plasma is exceedingly complex. Why does the chromatogram show just one clean peak?
(e) Given that -cocaine has only two major mass spectral peaks at m/z 309 and 182, which atoms are labelled with deuterium?
(f) Explain how you would use -cocaine for measuring cocaine in blood.
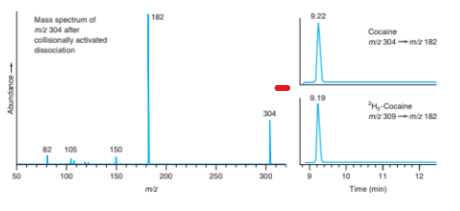
Spectrum for Problem 25-25.
Left: Mass spectrum of collisionally activated dissociation products from m/z 304 positive ion from atmospheric pressure chemical ionization mass spectrum of cocaine.
Right: Chromatograms obtained by selected reaction monitoring. [Data from G. Singh, V. Arora, P. T. Fenn, B. Mets, and I. A. Blair, “Isotope Dilution Liquid Chromatography Tandem Mass Spectrometry Assay for Trace Analysis of Cocaine and Its Metabolites in Plasma,” Anal. Chem. 1999, 71, 2021.]
Short Answer
The part (a), part (b), part (c), part (d), part (e), part (f) is
The given structure of cocaine in the problem has an m/z of 303, and is not charged
The dissociation of this substituent and the hydrogen atom would leave the original structure with a m/z of 182
The ions containing the isotopes, having a m/z of 305
The ions with the isotopes, the ions that did not have a m/z of 304 or m/z of 182
The 5 deuterium ions should be located in this fragment so that when it gets dissociated
-cocaine is determined and quantified into the response factor