Chapter 25: Q18P (page 708)
In monolithic columns60 the stationary phase is a single porous piece of silica or polymer filling the entire column and synthesized within the column from liquid precursors. Monolithic columns offer similar plate height to HPLC particles, but with less resistance to flow. Therefore, faster flow or longer columns can be used. The figure shows separation of isotopic molecules on a long monolithic column. Packed columns have too much resistance to flow to be made so long.
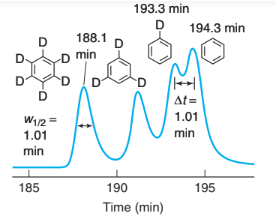
Separation of isotopic molecules on a 440-cm-long monolithic C18-silica column eluted with(30: 70 vol/vol) at 308C. [Data from K. Miyamoto, T. Hara, H. Kobayashi, H. Morisaka, D. Tokuda, K. Horie, K. Koduki, S. Makino, O. Nuñez, C. Yang, T. Kawabe, T. Ikegami, H. Takubo, Y. Ishihama, and N. Tanaka, “High-Efficiency Liquid Chromatographic Separation Utilizing Long Monolithic Silica Capillary Columns,” Anal. Chem. 2008, 80, 8741.]
(a) Unretained thiourea is eluted in 41.7 min. Find the linear velocity ux (mm/s).
(b) Find the retention factor k for
(c) Find the plate number N and plate height for
(d) Assuming that the peak widths forand are the same as that of , find the resolution of and .
(f) If we just increased the column length to increase N, what value of N and what column length would be required for a resolution of 1.000?
(g) Without increasing the length of the column, and without changing the stationary phase, how might you improve the resolution?
(h) When the solvent was changed from(30:70 vol/vol) to(10:5:85), the relative retention for C6H5D andincreased to 1.0088 and the retention factor for C6H6 changed to 17.0. If the plate number were unchanged, what would be the resolution?
The part (a), part (b), part (c), part (d), part (e), part (f), part (g), part (h) is
a)
b)
c)
d)
e)
f)
g) Decreasing the flow (decreases H and increases N) and decreasing mobile phase strength
h)
Step by step solution
01
Linear velocity ux (mm/s)
Part (a)
The linear velocity is calculated using the formula:
02
Retention factor k for C6D6
Part (b)
Formula for the retention factor is:
Retention factor for is:
03
Plate number N and plate height for C6D6
Part (c)
Formula for the plate number N is:
The N is:
The plate heigh is:
04
Resolution of C6H5D and C6 D6
Part (d)
The resolution can be calculated using the formula:
05
Relative retention (a) between C6H5D and C6D6
Part (e)
The relative retention is:
06
Value of N and column length
Part (f)
We need to calculate form formula for resolution:
Where the is:
And the is:
is:
Now we express N and put values in the formula to get N:
A column length of 440 cm gave N=192000. So, we must multiply length with factor:
The length is:
07
Improve the resolution
Part (g)
Without increasing the length of the column, and without changing the stationary phase, we might improve the resolution by changing the solvent, decreasing the flow (decreases H and increases N) and decreasing mobile phase strength.
08
The resolution
Part (h)
The resolution is:
Here, the result of part (a), part (b), part (c), part (d), part(e), part (f), part (g), part (h) is
(a)
(b)
(c)
(d)
(e)
(f)
(g) Decreasing the flow (decreases H and increases N) and decreasing mobile phase strength
(h)
Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their
learning with 91Ӱ��!




